| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 23:49:57 UTC |
|---|
| Update Date | 2016-11-09 01:18:29 UTC |
|---|
| Accession Number | CHEM027584 |
|---|
| Identification |
|---|
| Common Name | Casimiroin |
|---|
| Class | Small Molecule |
|---|
| Description | Casimiroin is a quinone reductase 2 inhibitor isolated from _Casimiroa edulis_. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
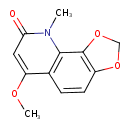
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 6-Methoxy-9-methyl-(1,3)dioxolo(4,5-H)quinolin-8(9H)-one | MeSH | | 4-Methoxy-1-methyl-7,8-methylenedioxy-2(1H)-quinolinone | HMDB | | 6-Methoxy-9-methyl-1,3-dioxolo(4,5-H)quinolin-8(9H)-one | HMDB | | 6-Methoxy-9-methyl-1,3-dioxolo[4,5-H]quinolin-8(9H)-one, 9ci | HMDB |
|
|---|
| Chemical Formula | C12H11NO4 |
|---|
| Average Molecular Mass | 233.220 g/mol |
|---|
| Monoisotopic Mass | 233.069 g/mol |
|---|
| CAS Registry Number | 477-89-4 |
|---|
| IUPAC Name | 6-methoxy-9-methyl-2H,8H,9H-[1,3]dioxolo[4,5-h]quinolin-8-one |
|---|
| Traditional Name | casimiroin |
|---|
| SMILES | COC1=CC(=O)N(C)C2=C1C=CC1=C2OCO1 |
|---|
| InChI Identifier | InChI=1S/C12H11NO4/c1-13-10(14)5-9(15-2)7-3-4-8-12(11(7)13)17-6-16-8/h3-5H,6H2,1-2H3 |
|---|
| InChI Key | DPXXJCMMMXZVSW-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as hydroquinolones. Hydroquinolones are compounds containing a hydrogenated quinoline bearing a ketone group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Quinolines and derivatives |
|---|
| Sub Class | Quinolones and derivatives |
|---|
| Direct Parent | Hydroquinolones |
|---|
| Alternative Parents | |
|---|
| Substituents | - Dihydroquinolone
- Dihydroquinoline
- Benzodioxole
- Alkyl aryl ether
- Pyridinone
- Pyridine
- Benzenoid
- Heteroaromatic compound
- Vinylogous ester
- Lactam
- Ether
- Oxacycle
- Azacycle
- Acetal
- Hydrocarbon derivative
- Organopnictogen compound
- Organic oxygen compound
- Organic oxide
- Organonitrogen compound
- Organooxygen compound
- Organic nitrogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0uxr-1590000000-c70b59dc35bdf60d7e50 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-0090000000-f042b42babccdf7dfe0c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-0090000000-b6335089477b8fda1dc8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-114l-1910000000-d8e1c63a6c4304964432 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-001i-0090000000-6422449a3590d6ceef75 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-001i-0090000000-fffc6bc47661e6265ef7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0uki-2970000000-f56873d8712144e595c2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-0090000000-a3c4ed76b69cb58b201e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-0090000000-7d2db07fd57fde671a3a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0ue9-0950000000-a775d84cd25f2d1ae31e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-001i-0090000000-ca3cfa1e5f9142eb756d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-001i-0190000000-4872775dd5aa62619e65 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0fka-0920000000-3c32cef7ab4d6eb2c3e7 | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB08744 |
|---|
| HMDB ID | HMDB0033349 |
|---|
| FooDB ID | FDB011377 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00002146 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 110572 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 124075 |
|---|
| Kegg Compound ID | C10654 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|