| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 23:47:06 UTC |
|---|
| Update Date | 2016-11-09 01:18:28 UTC |
|---|
| Accession Number | CHEM027506 |
|---|
| Identification |
|---|
| Common Name | Dihydroclusin |
|---|
| Class | Small Molecule |
|---|
| Description | Dihydroclusin is found in herbs and spices. Dihydroclusin is a constituent of Piper cubeba (cubeb pepper). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
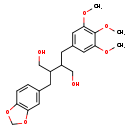
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 9,9'-Dihydroxy-3,4,5-trimethoxy-3',4'-methylenedioxylignan | HMDB | | Podophyllotoxin, secodeoxy secolactonediol | HMDB |
|
|---|
| Chemical Formula | C22H28O7 |
|---|
| Average Molecular Mass | 404.454 g/mol |
|---|
| Monoisotopic Mass | 404.184 g/mol |
|---|
| CAS Registry Number | 73149-51-6 |
|---|
| IUPAC Name | 2-(2H-1,3-benzodioxol-5-ylmethyl)-3-[(3,4,5-trimethoxyphenyl)methyl]butane-1,4-diol |
|---|
| Traditional Name | 2-(2H-1,3-benzodioxol-5-ylmethyl)-3-[(3,4,5-trimethoxyphenyl)methyl]butane-1,4-diol |
|---|
| SMILES | COC1=CC(CC(CO)C(CO)CC2=CC3=C(OCO3)C=C2)=CC(OC)=C1OC |
|---|
| InChI Identifier | InChI=1S/C22H28O7/c1-25-20-9-15(10-21(26-2)22(20)27-3)7-17(12-24)16(11-23)6-14-4-5-18-19(8-14)29-13-28-18/h4-5,8-10,16-17,23-24H,6-7,11-13H2,1-3H3 |
|---|
| InChI Key | FDHFWHRGVDRJIK-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as dibenzylbutanediol lignans. These are lignan compounds containing a 2,3-dibenzylbutane-1,4-diol moiety. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lignans, neolignans and related compounds |
|---|
| Class | Dibenzylbutane lignans |
|---|
| Sub Class | Dibenzylbutanediol lignans |
|---|
| Direct Parent | Dibenzylbutanediol lignans |
|---|
| Alternative Parents | |
|---|
| Substituents | - Dibenzylbutanediol
- Benzodioxole
- Phenoxy compound
- Methoxybenzene
- Anisole
- Phenol ether
- Alkyl aryl ether
- Benzenoid
- Monocyclic benzene moiety
- Acetal
- Oxacycle
- Organoheterocyclic compound
- Ether
- Organic oxygen compound
- Organooxygen compound
- Primary alcohol
- Hydrocarbon derivative
- Alcohol
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-00dr-0928000000-b283bf64fbdfc2ffe35c | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-001i-9730560000-ba50727fe4e9b58aa1c7 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-052r-0029300000-ca37a298eb3a3a5680df | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014r-0389000000-461ca3edf7226732690c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4i-0289000000-243f8f38815b4a7dad0b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0008900000-b5fe5efe7fbd3f165cd0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0pi9-0009100000-c15a67a7758a7058327f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a70-1249000000-d50e82ae2aebd8f3b193 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0319700000-6792938a97f7d269b40f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0ap0-0739100000-fc180620ccd1165bda32 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0api-0669000000-9bac2bf84063d950f0bf | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0004900000-e77ba05efe94da587769 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0pi3-0009100000-063e16a17890592bc6db | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-0459100000-da01bdb5f73436581960 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0033272 |
|---|
| FooDB ID | FDB011295 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00041986 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 3198127 |
|---|
| ChEBI ID | 175230 |
|---|
| PubChem Compound ID | 3978441 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|