| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 23:40:54 UTC |
|---|
| Update Date | 2016-11-09 01:18:26 UTC |
|---|
| Accession Number | CHEM027352 |
|---|
| Identification |
|---|
| Common Name | Ganoderic acid W |
|---|
| Class | Small Molecule |
|---|
| Description | Ganoderic acid W is found in mushrooms. Ganoderic acid W is a constituent of Ganoderma lucidum (reishi). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
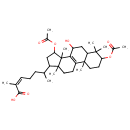
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Ganoderate W | Generator | | (2Z)-6-[5,12-Bis(acetyloxy)-9-hydroxy-2,6,6,11,15-pentamethyltetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadec-1(10)-en-14-yl]-2-methylhept-2-enoate | HMDB |
|
|---|
| Chemical Formula | C34H52O7 |
|---|
| Average Molecular Mass | 572.773 g/mol |
|---|
| Monoisotopic Mass | 572.371 g/mol |
|---|
| CAS Registry Number | 86377-49-3 |
|---|
| IUPAC Name | (2Z)-6-[5,12-bis(acetyloxy)-9-hydroxy-2,6,6,11,15-pentamethyltetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadec-1(10)-en-14-yl]-2-methylhept-2-enoic acid |
|---|
| Traditional Name | (2Z)-6-[5,12-bis(acetyloxy)-9-hydroxy-2,6,6,11,15-pentamethyltetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadec-1(10)-en-14-yl]-2-methylhept-2-enoic acid |
|---|
| SMILES | CC(CC\C=C(\C)C(O)=O)C1CC(OC(C)=O)C2(C)C3=C(CCC12C)C1(C)CCC(OC(C)=O)C(C)(C)C1CC3O |
|---|
| InChI Identifier | InChI=1S/C34H52O7/c1-19(11-10-12-20(2)30(38)39)24-17-28(41-22(4)36)34(9)29-23(13-16-33(24,34)8)32(7)15-14-27(40-21(3)35)31(5,6)26(32)18-25(29)37/h12,19,24-28,37H,10-11,13-18H2,1-9H3,(H,38,39)/b20-12- |
|---|
| InChI Key | DIXZQXARJMRUPX-NDENLUEZSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as triterpenoids. These are terpene molecules containing six isoprene units. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Triterpenoids |
|---|
| Direct Parent | Triterpenoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Triterpenoid
- Hydroxy bile acid, alcohol, or derivatives
- Monohydroxy bile acid, alcohol, or derivatives
- Bile acid, alcohol, or derivatives
- Steroid ester
- Steroid acid
- Hydroxysteroid
- 7-hydroxysteroid
- Steroid
- Tricarboxylic acid or derivatives
- Carboxylic acid ester
- Secondary alcohol
- Carboxylic acid
- Carboxylic acid derivative
- Hydrocarbon derivative
- Organic oxide
- Alcohol
- Carbonyl group
- Organic oxygen compound
- Organooxygen compound
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-06tr-0101950000-4b516c35959959016b29 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-00c0-2011194000-6aaf680560f13684eef5 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS ("Ganoderic acid W,1TMS,#1" TMS) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_2) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_2) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_2_1) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0c0r-0000290000-7b812065cae2caa7a962 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-08fs-0000980000-d9d6448a7d132577d127 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0159-1101920000-9adcb79714f5b79c0d23 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-024i-0000090000-bc1fecbd2bfe9d850898 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-06vi-2000390000-903b1594c25178b51f10 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-052r-6000940000-71eb2bde9f215de84f62 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00dj-4003980000-0ead7831aca2a1e261f0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0005-9000520000-e45449e35dbd7df86bdf | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-014l-9004200000-79706c76e85f760a6046 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-3000090000-52fe267e83524c7bae53 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0ar0-6000890000-c0aa86eb4b0a7be8a228 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0005-9000120000-52ced0fc935f49e6312e | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0033078 |
|---|
| FooDB ID | FDB011073 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 74886390 |
|---|
| ChEBI ID | 176117 |
|---|
| PubChem Compound ID | 131751368 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|