| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 23:33:52 UTC |
|---|
| Update Date | 2016-11-09 01:18:24 UTC |
|---|
| Accession Number | CHEM027194 |
|---|
| Identification |
|---|
| Common Name | Amaranth |
|---|
| Class | Small Molecule |
|---|
| Description | Amaranthus, collectively known as amaranth, is a cosmopolitan genus of annual or short-lived perennial plants. Catkin-like cymes of densely packed flowers are borne in summer or autumn. Approximately 60 species are recognized, with inflorescences and foliage ranging from purple and red to green or gold. Members of this genus share many characteristics and uses with members of the closely related genus Celosia. Several species are raised for amaranth "grain" in Asia and the Americas. Its seeds are a good source of protein. Compared to other grains, amaranth is unusually rich in the essential amino acid lysine. Common grains such as wheat and corn are comparatively rich in amino acids that amaranth lacks; thus, amaranth and other grains can complement each other.Cooked amaranth leaves are a good source of vitamin A, vitamin C, and folate; they are also a complementing source of other vitamins such as thiamine, niacin, and riboflavin, plus some dietary minerals including calcium, iron, potassium, zinc, copper, and manganese. Cooked amaranth grains are a complementing source of thiamine, niacin, riboflavin, and folate, and dietary minerals including calcium, iron, magnesium, phosphorus, zinc, copper, and manganese - comparable to common grains such as wheat germ, oats and others.Amaranth may be a promising source of protein to those who are gluten sensitive, because unlike the protein found in grains such as wheat and rye, its protein does not contain gluten. According to a 2007 report, amaranth compares well in nutrient content with gluten-free vegetarian options such as buckwheat, corn, millet, wild rice, oats and quinoa. (Wikipedia). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
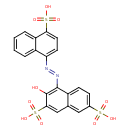
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3-Hydroxy-4-[(4-sulfO-1-naphthalenyl)azo]-2,7-naphthalenedisulfonic acid, 9ci | HMDB | | Amaranth R | HMDB | | Amaranth S | HMDB | | Bordeaux S | HMDB | | C.I. 16185 | HMDB | | C.I. acid red 27, 8ci | HMDB | | C.I. FOOD red 9 | HMDB | | e123 | HMDB | | FD And C red no. 2 | HMDB | | 3-Hydroxy-4-[(e)-2-(4-sulfonaphthalen-1-yl)diazen-1-yl]naphthalene-2,7-disulfonate | Generator | | 3-Hydroxy-4-[(e)-2-(4-sulphonaphthalen-1-yl)diazen-1-yl]naphthalene-2,7-disulphonate | Generator | | 3-Hydroxy-4-[(e)-2-(4-sulphonaphthalen-1-yl)diazen-1-yl]naphthalene-2,7-disulphonic acid | Generator |
|
|---|
| Chemical Formula | C20H14N2O10S3 |
|---|
| Average Molecular Mass | 538.528 g/mol |
|---|
| Monoisotopic Mass | 537.981 g/mol |
|---|
| CAS Registry Number | 642-59-1 |
|---|
| IUPAC Name | 3-hydroxy-4-[(E)-2-(4-sulfonaphthalen-1-yl)diazen-1-yl]naphthalene-2,7-disulfonic acid |
|---|
| Traditional Name | 3-hydroxy-4-[(E)-2-(4-sulfonaphthalen-1-yl)diazen-1-yl]naphthalene-2,7-disulfonic acid |
|---|
| SMILES | OC1=C(\N=N\C2=CC=C(C3=CC=CC=C23)S(O)(=O)=O)C2=CC=C(C=C2C=C1S(O)(=O)=O)S(O)(=O)=O |
|---|
| InChI Identifier | InChI=1S/C20H14N2O10S3/c23-20-18(35(30,31)32)10-11-9-12(33(24,25)26)5-6-13(11)19(20)22-21-16-7-8-17(34(27,28)29)15-4-2-1-3-14(15)16/h1-10,23H,(H,24,25,26)(H,27,28,29)(H,30,31,32)/b22-21+ |
|---|
| InChI Key | IRPXADUBAQAOKL-QURGRASLSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as 1,1'-azonaphthalenes. These are organonitrogen aromatic compounds that contain a central azo group, where each nitrogen atom is conjugated to the 1-position of a naphthalene ring system. Naphthalene is a compound made up of two fused benzene rings. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Naphthalenes |
|---|
| Sub Class | 1,1'-azonaphthalenes |
|---|
| Direct Parent | 1,1'-azonaphthalenes |
|---|
| Alternative Parents | |
|---|
| Substituents | - 1,1'-azonaphthalene
- 2-naphthalene sulfonate
- 1-naphthalene sulfonate
- Naphthalene sulfonic acid or derivatives
- 2-naphthalene sulfonic acid or derivatives
- 1-naphthalene sulfonic acid or derivatives
- Naphthalene sulfonate
- 2-naphthol
- Arylsulfonic acid or derivatives
- 1-sulfo,2-unsubstituted aromatic compound
- Organic sulfonic acid or derivatives
- Organosulfonic acid or derivatives
- Organosulfonic acid
- Sulfonyl
- Azo compound
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Hydrocarbon derivative
- Organic oxide
- Organosulfur compound
- Organooxygen compound
- Organonitrogen compound
- Organopnictogen compound
- Organic oxygen compound
- Organic nitrogen compound
- Aromatic homopolycyclic compound
|
|---|
| Molecular Framework | Aromatic homopolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0abi-0192630000-17f8c82f944218592e21 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-00di-2042290000-c59bda7f4f73b9457925 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_2) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_3) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_4) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_2) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_3) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_4) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS ("Amaranth,1TMS,#1" TMS) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0079-0001290000-fbbc131a46401e4108f4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-0012930000-cacb2a64c53048caa40f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-004l-0559300000-ab882aef588e6ea8469a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0022190000-55bdfbe7499b6f9aaedb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-05tr-3175970000-5f0ee5004f2a80420e27 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0083-2391000000-7fa0c11859c7fb4f2e54 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0079-0000190000-7791a626e1be6e9c5314 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-000i-0197480000-067a19802072fc2b8ca6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a70-0984100000-d9584389f4100a51b72e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-1000090000-230ab5e60e5c5329084f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-001r-7000190000-ec01f9ffc1ebfadb5f46 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0059-8439200000-18da63d7dbf8ef27a976 | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0032885 |
|---|
| FooDB ID | FDB010867 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00000356 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Amaranth |
|---|
| Chemspider ID | 11200994 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|