| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 23:32:18 UTC |
|---|
| Update Date | 2016-11-09 01:18:24 UTC |
|---|
| Accession Number | CHEM027157 |
|---|
| Identification |
|---|
| Common Name | 3-Benzoyloxy-6-oxo-12-ursen-28-oic acid |
|---|
| Class | Small Molecule |
|---|
| Description | 3-Benzoyloxy-6-oxo-12-ursen-28-oic acid is found in fruits. 3-Benzoyloxy-6-oxo-12-ursen-28-oic acid is a constituent of the famine food Momordica dioica. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
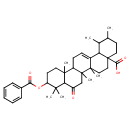
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3-Benzoyloxy-6-oxo-12-ursen-28-Oate | Generator | | 10-(Benzoyloxy)-1,2,6a,6b,9,9,12a-heptamethyl-8-oxo-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,12b,13,14b-icosahydropicene-4a-carboxylate | HMDB |
|
|---|
| Chemical Formula | C37H50O5 |
|---|
| Average Molecular Mass | 574.790 g/mol |
|---|
| Monoisotopic Mass | 574.366 g/mol |
|---|
| CAS Registry Number | 205699-24-7 |
|---|
| IUPAC Name | 10-(benzoyloxy)-1,2,6a,6b,9,9,12a-heptamethyl-8-oxo-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,12b,13,14b-icosahydropicene-4a-carboxylic acid |
|---|
| Traditional Name | 10-(benzoyloxy)-1,2,6a,6b,9,9,12a-heptamethyl-8-oxo-1,2,3,4,5,6,7,8a,10,11,12,12b,13,14b-tetradecahydropicene-4a-carboxylic acid |
|---|
| SMILES | CC1CCC2(CCC3(C)C(=CCC4C5(C)CCC(OC(=O)C6=CC=CC=C6)C(C)(C)C5C(=O)CC34C)C2C1C)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C37H50O5/c1-22-15-18-37(32(40)41)20-19-35(6)25(29(37)23(22)2)13-14-27-34(5)17-16-28(42-31(39)24-11-9-8-10-12-24)33(3,4)30(34)26(38)21-36(27,35)7/h8-13,22-23,27-30H,14-21H2,1-7H3,(H,40,41) |
|---|
| InChI Key | WTRSURIFLHBSLZ-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as triterpenoids. These are terpene molecules containing six isoprene units. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Triterpenoids |
|---|
| Direct Parent | Triterpenoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Triterpenoid
- Benzoate ester
- Benzoic acid or derivatives
- Benzoyl
- Dicarboxylic acid or derivatives
- Monocyclic benzene moiety
- Benzenoid
- Ketone
- Carboxylic acid ester
- Carboxylic acid
- Carboxylic acid derivative
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Organic oxide
- Organic oxygen compound
- Aromatic homopolycyclic compound
|
|---|
| Molecular Framework | Aromatic homopolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0a4i-3332490000-f818e3bdee0f346e1def | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-053r-3311139000-fd15186dee49d113569d | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS ("3-Benzoyloxy-6-oxo-12-ursen-28-oic acid,1TMS,#1" TMS) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_2) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_3) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_2) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_3) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-056r-0200290000-7b7f379cf4a5a6859f76 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-0700890000-19123781290785308435 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4i-4904420000-b7910b42eceddccc5174 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-0000190000-a689cec1b2034d583aeb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00b9-2300390000-638ad7c5e588f812550e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0fi0-6600910000-c3dbb891efd9c4032fd8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0pdi-0100960000-2b127473e7d274384f7c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-0222940000-29691def6c7f505b313e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0fki-3953100000-afe508597e46c461d43c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-0000090000-09460550243255eea2c3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00di-2200190000-299b45995bc2c7fb5474 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00b9-8300090000-f964da7e2aa5ae484cbf | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0032840 |
|---|
| FooDB ID | FDB010817 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00055197 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 35013503 |
|---|
| ChEBI ID | 172749 |
|---|
| PubChem Compound ID | 131751331 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|