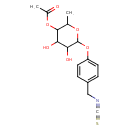| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 23:30:48 UTC |
|---|
| Update Date | 2016-11-09 01:18:24 UTC |
|---|
| Accession Number | CHEM027124 |
|---|
| Identification |
|---|
| Common Name | 4-Hydroxybenzyl isothiocyanate 4''-acetylrhamnoside |
|---|
| Class | Small Molecule |
|---|
| Description | Constituent of seeds of Moringa oleifera (horseradish tree) and Moringa peregrina. 4-Hydroxybenzyl isothiocyanate 4''-acetylrhamnoside is found in herbs and spices. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 4-Hydroxybenzyl isothiocyanic acid 4''-acetylrhamnoside | Generator | | 4,5-Dihydroxy-6-[4-(isothiocyanatomethyl)phenoxy]-2-methyloxan-3-yl acetic acid | Generator |
|
|---|
| Chemical Formula | C16H19NO6S |
|---|
| Average Molecular Mass | 353.390 g/mol |
|---|
| Monoisotopic Mass | 353.093 g/mol |
|---|
| CAS Registry Number | 73255-41-1 |
|---|
| IUPAC Name | 4,5-dihydroxy-6-[4-(isothiocyanatomethyl)phenoxy]-2-methyloxan-3-yl acetate |
|---|
| Traditional Name | 4,5-dihydroxy-6-[4-(isothiocyanatomethyl)phenoxy]-2-methyloxan-3-yl acetate |
|---|
| SMILES | CC1OC(OC2=CC=C(CN=C=S)C=C2)C(O)C(O)C1OC(C)=O |
|---|
| InChI Identifier | InChI=1S/C16H19NO6S/c1-9-15(22-10(2)18)13(19)14(20)16(21-9)23-12-5-3-11(4-6-12)7-17-8-24/h3-6,9,13-16,19-20H,7H2,1-2H3 |
|---|
| InChI Key | WVUZLAMBBZISKM-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as phenolic glycosides. These are organic compounds containing a phenolic structure attached to a glycosyl moiety. Some examples of phenolic structures include lignans, and flavonoids. Among the sugar units found in natural glycosides are D-glucose, L-Fructose, and L rhamnose. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbohydrates and carbohydrate conjugates |
|---|
| Direct Parent | Phenolic glycosides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Phenolic glycoside
- Hexose monosaccharide
- O-glycosyl compound
- Phenoxy compound
- Phenol ether
- Monocyclic benzene moiety
- Monosaccharide
- Oxane
- Benzenoid
- 1,2-diol
- Carboxylic acid ester
- Isothiocyanate
- Secondary alcohol
- Acetal
- Carboxylic acid derivative
- Oxacycle
- Monocarboxylic acid or derivatives
- Organoheterocyclic compound
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Hydrocarbon derivative
- Organic oxide
- Alcohol
- Organopnictogen compound
- Carbonyl group
- Organonitrogen compound
- Organosulfur compound
- Organic nitrogen compound
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-052f-8982000000-ae79eed67c4258207283 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-053u-4841900000-a77bf3f0b021d809dbb3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0945000000-3a911f4c2366f833785a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-066r-0910000000-3344216fce776861d7ca | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4i-1900000000-e4fc079a428e372a509d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0ik9-6719000000-e0ff89e245928d392037 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-08fr-6912000000-dcab7c8edb82c956aa43 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-9400000000-5949e408a342d5037d11 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0032802 |
|---|
| FooDB ID | FDB010774 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 22718840 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|