| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 23:27:19 UTC |
|---|
| Update Date | 2016-11-09 01:18:23 UTC |
|---|
| Accession Number | CHEM027036 |
|---|
| Identification |
|---|
| Common Name | Bis(2-methylpropanoyloxy)-9,10-epoxy-p-mentha-1,3,5-triene |
|---|
| Class | Small Molecule |
|---|
| Description | Bis(2-methylpropanoyloxy)-9,10-epoxy-p-mentha-1,3,5-triene is found in fats and oils. Bis(2-methylpropanoyloxy)-9,10-epoxy-p-mentha-1,3,5-triene is a constituent of various plant species including Madia sativa (Chile tarweed) |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
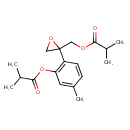
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Mestranol bicarbonate | HMDB | | (2-{4-methyl-2-[(2-methylpropanoyl)oxy]phenyl}oxiran-2-yl)methyl 2-methylpropanoic acid | HMDB |
|
|---|
| Chemical Formula | C18H24O5 |
|---|
| Average Molecular Mass | 320.380 g/mol |
|---|
| Monoisotopic Mass | 320.162 g/mol |
|---|
| CAS Registry Number | 22518-06-5 |
|---|
| IUPAC Name | (2-{4-methyl-2-[(2-methylpropanoyl)oxy]phenyl}oxiran-2-yl)methyl 2-methylpropanoate |
|---|
| Traditional Name | (2-{4-methyl-2-[(2-methylpropanoyl)oxy]phenyl}oxiran-2-yl)methyl 2-methylpropanoate |
|---|
| SMILES | CC(C)C(=O)OCC1(CO1)C1=C(OC(=O)C(C)C)C=C(C)C=C1 |
|---|
| InChI Identifier | InChI=1S/C18H24O5/c1-11(2)16(19)21-9-18(10-22-18)14-7-6-13(5)8-15(14)23-17(20)12(3)4/h6-8,11-12H,9-10H2,1-5H3 |
|---|
| InChI Key | OLARKEMZPWGFJU-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as phenol esters. These are aromatic compounds containing a benzene ring substituted by a hydroxyl group and an ester group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Phenol esters |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Phenol esters |
|---|
| Alternative Parents | |
|---|
| Substituents | - Phenol ester
- Phenoxy compound
- Toluene
- Monocyclic benzene moiety
- Dicarboxylic acid or derivatives
- Carboxylic acid ester
- Carboxylic acid derivative
- Dialkyl ether
- Oxirane
- Ether
- Oxacycle
- Organoheterocyclic compound
- Hydrocarbon derivative
- Carbonyl group
- Organooxygen compound
- Organic oxide
- Organic oxygen compound
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-00r5-8790000000-fd1cf3e58db53737df05 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00e9-6198000000-b15d9f9ff69bbb380648 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0089-9371000000-aaa69914f493093f8c3b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-05fu-9000000000-b8d58cedfb180a6154ef | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014l-7639000000-49f0dc7638ade490fbbd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-014r-9233000000-ae43c89dbc0ecc9d5296 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-05pc-9800000000-7ce0888ba4c036d53be5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00e9-0269000000-7a87148e43d9b205c077 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-2592000000-9ed48aba8e6aed99d47d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-7941000000-b8d8ca8c98c849cbe6f0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-002k-4980000000-48a3460c98f1cc9ac422 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0079-9310000000-6f5f745b93ba3cf8a8a0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00ej-9510000000-3900ee635e8bde230521 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0032698 |
|---|
| FooDB ID | FDB010655 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 9647499 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 11472669 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|