| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 23:26:22 UTC |
|---|
| Update Date | 2016-11-09 01:18:22 UTC |
|---|
| Accession Number | CHEM027011 |
|---|
| Identification |
|---|
| Common Name | (E)-2',4,4'-Trihydroxy-3-prenylchalcone |
|---|
| Class | Small Molecule |
|---|
| Description | (E)-2',4,4'-Trihydroxy-3-prenylchalcone is found in herbs and spices. (E)-2',4,4'-Trihydroxy-3-prenylchalcone is a constituent of hairy root cultures of Glycyrrhiza glabra (licorice) |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
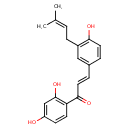
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3-Prenyl-4,2',4'-trihydroxychalcone | HMDB | | Licoagrochalcone a | HMDB |
|
|---|
| Chemical Formula | C20H20O4 |
|---|
| Average Molecular Mass | 324.370 g/mol |
|---|
| Monoisotopic Mass | 324.136 g/mol |
|---|
| CAS Registry Number | 202815-28-9 |
|---|
| IUPAC Name | (2E)-1-(2,4-dihydroxyphenyl)-3-[4-hydroxy-3-(3-methylbut-2-en-1-yl)phenyl]prop-2-en-1-one |
|---|
| Traditional Name | licoagrochalcone A |
|---|
| SMILES | CC(C)=CCC1=C(O)C=CC(\C=C\C(=O)C2=C(O)C=C(O)C=C2)=C1 |
|---|
| InChI Identifier | InChI=1S/C20H20O4/c1-13(2)3-6-15-11-14(4-9-18(15)22)5-10-19(23)17-8-7-16(21)12-20(17)24/h3-5,7-12,21-22,24H,6H2,1-2H3/b10-5+ |
|---|
| InChI Key | TVUGLERLRIQATC-BJMVGYQFSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as 2'-hydroxychalcones. These are organic compounds containing chalcone skeleton that carries a hydroxyl group at the 2'-position. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Linear 1,3-diarylpropanoids |
|---|
| Sub Class | Chalcones and dihydrochalcones |
|---|
| Direct Parent | 2'-Hydroxychalcones |
|---|
| Alternative Parents | |
|---|
| Substituents | - 2'-hydroxychalcone
- Cinnamylphenol
- Hydroxycinnamic acid or derivatives
- Benzoyl
- Resorcinol
- Styrene
- Aryl ketone
- Phenol
- 1-hydroxy-4-unsubstituted benzenoid
- 1-hydroxy-2-unsubstituted benzenoid
- Monocyclic benzene moiety
- Benzenoid
- Vinylogous acid
- Acryloyl-group
- Enone
- Alpha,beta-unsaturated ketone
- Ketone
- Hydrocarbon derivative
- Organooxygen compound
- Organic oxide
- Organic oxygen compound
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-052u-5986000000-dad20441f13bffb65829 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-004i-3010490000-11362303b8f460b2d203 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-0339000000-e9a964a16d50bb457e25 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01bi-2942000000-c14e533d30e71310cb0d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-014r-6910000000-6858546c066653302c2e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-0209000000-e2c93bb5f886a438059d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00di-0639000000-fc0f34548ea4770e9880 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-5930000000-d9e76381c8a6b0682b02 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-0009000000-b64caba2598a2db6662e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00dr-0829000000-f2fbcee6cef9c8eb48a3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-01c9-1930000000-7ca5855ccf234de3a209 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0292000000-5b1c2d7153bcd6de4d7c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4j-1961000000-c49e549086fa7457360a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-017u-2940000000-2a68475ec97ab2a815a5 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0032671 |
|---|
| FooDB ID | FDB010625 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00014456 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 9274517 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 11099375 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|