| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 23:24:00 UTC |
|---|
| Update Date | 2016-11-09 01:18:22 UTC |
|---|
| Accession Number | CHEM026960 |
|---|
| Identification |
|---|
| Common Name | Sodium glycocholate |
|---|
| Class | Small Molecule |
|---|
| Description | Glycocholic acid, also known as glycocholate or N-choloyl-glycine, belongs to the class of organic compounds known as glycinated bile acids and derivatives. Glycinated bile acids and derivatives are compounds with a structure characterized by the presence of a glycine linked to a bile acid skeleton. Glycocholic acid exists as a solid, possibly soluble (in water), and an extremely weak basic (essentially neutral) compound (based on its pKa) molecule. Glycocholic acid has been found to be associated with several diseases known as crohn's disease, hepatobiliary diseases, cystic fibrosis, and portal vein obstruction; also glycocholic acid has been linked to the inborn metabolic disorders including celiac disease. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
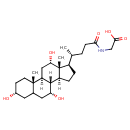
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Glycocholate | Generator | | 3a,7a,12a-Trihydroxy-5b-cholan-24-oylglycine | HMDB | | 3a,7a,12a-Trihydroxy-5b-cholanic acid-24-glycine | HMDB | | 3a,7a,12a-Trihydroxy-N-(carboxymethyl)-5b-cholan-24-amide | HMDB | | Cholylglycine | HMDB | | Glycine cholate | HMDB | | Glycoreductodehydrocholic acid | HMDB | | Glycylcholate | HMDB | | Glycylcholic acid | HMDB | | N-(Carboxymethyl)-3a,7a,12a-trihydroxy-5b-cholan-24-amide | HMDB | | N-Choloyl-glycine | HMDB | | N-Choloylglycine | HMDB | | Glycocholic acid, sodium salt | MeSH, HMDB | | Glycocholate sodium | MeSH, HMDB |
|
|---|
| Chemical Formula | C26H43NO6 |
|---|
| Average Molecular Mass | 465.623 g/mol |
|---|
| Monoisotopic Mass | 465.309 g/mol |
|---|
| CAS Registry Number | 863-57-0 |
|---|
| IUPAC Name | 2-[(4R)-4-[(1S,2S,5R,9R,10R,11S,14R,15R,16S)-5,9,16-trihydroxy-2,15-dimethyltetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadecan-14-yl]pentanamido]acetic acid |
|---|
| Traditional Name | [(4R)-4-[(1S,2S,5R,9R,10R,11S,14R,15R,16S)-5,9,16-trihydroxy-2,15-dimethyltetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadecan-14-yl]pentanamido]acetic acid |
|---|
| SMILES | CC(CCC(=O)NCC(O)=O)C1CCC2C3C(O)CC4CC(O)CCC4(C)C3CC(O)C12C |
|---|
| InChI Identifier | InChI=1S/C26H43NO6/c1-14(4-7-22(31)27-13-23(32)33)17-5-6-18-24-19(12-21(30)26(17,18)3)25(2)9-8-16(28)10-15(25)11-20(24)29/h14-21,24,28-30H,4-13H2,1-3H3,(H,27,31)(H,32,33) |
|---|
| InChI Key | RFDAIACWWDREDC-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as glycinated bile acids and derivatives. Glycinated bile acids and derivatives are compounds with a structure characterized by the presence of a glycine linked to a bile acid skeleton. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Steroids and steroid derivatives |
|---|
| Sub Class | Bile acids, alcohols and derivatives |
|---|
| Direct Parent | Glycinated bile acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Glycinated bile acid
- Trihydroxy bile acid, alcohol, or derivatives
- Hydroxy bile acid, alcohol, or derivatives
- 3-hydroxysteroid
- 12-hydroxysteroid
- Hydroxysteroid
- 7-hydroxysteroid
- 3-alpha-hydroxysteroid
- N-acyl-alpha-amino acid
- N-acyl-alpha amino acid or derivatives
- Alpha-amino acid or derivatives
- Fatty acyl
- N-acyl-amine
- Fatty amide
- Cyclic alcohol
- Secondary alcohol
- Secondary carboxylic acid amide
- Carboxamide group
- Polyol
- Monocarboxylic acid or derivatives
- Carboxylic acid derivative
- Carboxylic acid
- Organooxygen compound
- Organic oxygen compound
- Organopnictogen compound
- Organic oxide
- Hydrocarbon derivative
- Organic nitrogen compound
- Carbonyl group
- Alcohol
- Organonitrogen compound
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-00ds-0242900000-e424cae36874d8c9b014 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-01b9-5111349000-e192ab11f20a4e01e521 | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Negative (Annotated) | splash10-03di-0000900000-52e93813a9681b93b67c | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Negative (Annotated) | splash10-03k9-4000900000-79dfcaf6e620e89a8f51 | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Negative (Annotated) | splash10-00di-9000000000-0153bf31bdc096136252 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000t-1001900000-074a6139849914cbda16 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-057j-9004800000-a7c2f5dbba8bf1c9944b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-004i-9104300000-1463dae6a363a87b8243 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03dj-0000900000-885c6e5d3f15b31df656 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0h2b-3000900000-5c0d0e75172825307c79 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-05fr-9001000000-6241edfe03f378c4ba17 | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 2D NMR | [1H,13C] 2D NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0032596 |
|---|
| FooDB ID | FDB012346 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Glycocholic acid |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 23617285 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | | 1. Cortese, Frank; Bauman, Louis. A synthesis of conjugated bile acids. I. Glycocholic acid. Journal of the American Chemical Society (1935), 57 1393-5. | | 2. Qian L, Zhao A, Zhang Y, Chen T, Zeisel SH, Jia W, Cai W: Metabolomic Approaches to Explore Chemical Diversity of Human Breast-Milk, Formula Milk and Bovine Milk. Int J Mol Sci. 2016 Dec 17;17(12). pii: ijms17122128. doi: 10.3390/ijms17122128. |
|
|---|