| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 23:17:27 UTC |
|---|
| Update Date | 2016-11-09 01:18:21 UTC |
|---|
| Accession Number | CHEM026862 |
|---|
| Identification |
|---|
| Common Name | Rosmic acid |
|---|
| Class | Small Molecule |
|---|
| Description | An organic disulfide that results from the formal oxidative dimerisation of benzyl thiol. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
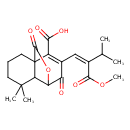
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 1,1'-[Dithiobis(methylene)]dibenzene | ChEBI | | 1,4-Diphenyl-2,3-dithiabutane | ChEBI | | 1,4-Diphenyl-2,3-dithiobutane | ChEBI | | alpha-(Benzyldithio)toluene | ChEBI | | BDS | ChEBI | | Benzyl bisulfide | ChEBI | | Benzyl disulfide | ChEBI | | Bis(phenylmethyl) disulfide | ChEBI | | Di(phenylmethyl) disulfide | ChEBI | | Dibenzyl disulphide | ChEBI | | a-(Benzyldithio)toluene | Generator | | Α-(benzyldithio)toluene | Generator | | Benzyl bisulphide | Generator | | Benzyl disulphide | Generator | | Bis(phenylmethyl) disulphide | Generator | | Di(phenylmethyl) disulphide | Generator | | 4,4'-Biphenyldiglyoxal disodium bisulfite | HMDB | | Aliphatic disulfide analog | HMDB | | Benzyl disulfide (8ci) | HMDB | | Benzyl disulfide, 8ci | HMDB | | Benzyldisulfanyl-methyl-benzene | HMDB | | Benzyldisulfide | HMDB | | Bis(phenylmethyl) disulfide, 9ci | HMDB | | Di(phenylmethyl)disulfide | HMDB | | Dibenzyldisulfid | HMDB | | Diphenylmethyl disulfide | HMDB | | Disulfide, bis(phenylmethyl) | HMDB | | Disulfide, dibenzyl | HMDB | | FEMA 3617 | HMDB | | Ghl.PD_Mitscher_leg0.312 | HMDB | | [(Benzyldisulfanyl)methyl]benzene | HMDB | | 9-[(1Z)-3-Methoxy-3-oxo-2-(propan-2-yl)prop-1-en-1-yl]-5,5-dimethyl-8,11-dioxo-12-oxatricyclo[5.3.2.0¹,⁶]dodec-9-ene-10-carboxylate | Generator | | Rosmate | Generator |
|
|---|
| Chemical Formula | C21H26O7 |
|---|
| Average Molecular Mass | 390.427 g/mol |
|---|
| Monoisotopic Mass | 390.168 g/mol |
|---|
| CAS Registry Number | 197799-63-6 |
|---|
| IUPAC Name | 9-[(1Z)-3-methoxy-3-oxo-2-(propan-2-yl)prop-1-en-1-yl]-5,5-dimethyl-8,11-dioxo-12-oxatricyclo[5.3.2.0¹,⁶]dodec-9-ene-10-carboxylic acid |
|---|
| Traditional Name | 9-[(1Z)-2-isopropyl-3-methoxy-3-oxoprop-1-en-1-yl]-5,5-dimethyl-8,11-dioxo-12-oxatricyclo[5.3.2.0¹,⁶]dodec-9-ene-10-carboxylic acid |
|---|
| SMILES | COC(=O)C(=C/C1=C(C(O)=O)C23CCCC(C)(C)C2C(OC3=O)C1=O)\C(C)C |
|---|
| InChI Identifier | InChI=1S/C21H26O7/c1-10(2)11(18(25)27-5)9-12-13(17(23)24)21-8-6-7-20(3,4)16(21)15(14(12)22)28-19(21)26/h9-10,15-16H,6-8H2,1-5H3,(H,23,24)/b11-9- |
|---|
| InChI Key | CAXSJVGHYYBJKT-LUAWRHEFSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as benzene and substituted derivatives. These are aromatic compounds containing one monocyclic ring system consisting of benzene. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Benzene and substituted derivatives |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Benzene and substituted derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Monocyclic benzene moiety
- Dialkyldisulfide
- Organic disulfide
- Sulfenyl compound
- Hydrocarbon derivative
- Organosulfur compound
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-001j-6109000000-a43ef3eb993eb5c09f39 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-0gis-7031900000-5f4a72285b2d35ce2966 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0009000000-e196235b2f8f27375905 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0006-3039000000-94b811684f0fb598faea | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00li-6493000000-b352914ef6bebf44870b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0009000000-d854cd5a580e2e1b8307 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-01rb-0139000000-36eb200374d6f856b141 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-014i-3596000000-02b0f4bb88b69ec44b9d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-01p9-0009000000-030e6b6608488fe7db95 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0019-0039000000-ec394c7f5854cc238a96 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-02w9-1779000000-e93995110e46e756266b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-052f-0019000000-4e561f2742fbd1618f3f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00kf-0429000000-2eed570d9eaebae61cb8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-03xu-5944000000-d82a0fcfdd477b3ff66a | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0032077 |
|---|
| FooDB ID | FDB008792 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00037046 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 8662 |
|---|
| ChEBI ID | 72752 |
|---|
| PubChem Compound ID | 9012 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|