| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 23:16:33 UTC |
|---|
| Update Date | 2016-11-09 01:18:20 UTC |
|---|
| Accession Number | CHEM026847 |
|---|
| Identification |
|---|
| Common Name | 2,4,14-Eicosatrienoic acid isobutylamide |
|---|
| Class | Small Molecule |
|---|
| Description | 2,4,14-Eicosatrienoic acid isobutylamide is an alkaloid from Piper retrofractum (Javanese long pepper |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
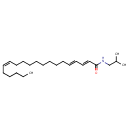
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 2,4,14-Eicosatrienoate isobutylamide | Generator | | N-Isobutyl-2,4,14-eicosatrienamide | HMDB | | (2E,4E,14Z)-N-(2-Methylpropyl)icosa-2,4,14-trienimidate | Generator |
|
|---|
| Chemical Formula | C24H43NO |
|---|
| Average Molecular Mass | 361.604 g/mol |
|---|
| Monoisotopic Mass | 361.334 g/mol |
|---|
| CAS Registry Number | 151391-70-7 |
|---|
| IUPAC Name | (2E,4E,14Z)-N-(2-methylpropyl)icosa-2,4,14-trienamide |
|---|
| Traditional Name | (2E,4E,14Z)-N-(2-methylpropyl)icosa-2,4,14-trienamide |
|---|
| SMILES | CCCCC\C=C/CCCCCCCC\C=C\C=C\C(=O)NCC(C)C |
|---|
| InChI Identifier | InChI=1S/C24H43NO/c1-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18-19-20-21-24(26)25-22-23(2)3/h8-9,18-21,23H,4-7,10-17,22H2,1-3H3,(H,25,26)/b9-8-,19-18+,21-20+ |
|---|
| InChI Key | GQCWFFNZERNJJC-SEXMCKGYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as n-acyl amines. N-acyl amines are compounds containing a fatty acid moiety linked to an amine group through an ester linkage. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Fatty Acyls |
|---|
| Sub Class | Fatty amides |
|---|
| Direct Parent | N-acyl amines |
|---|
| Alternative Parents | |
|---|
| Substituents | - N-acyl-amine
- Secondary carboxylic acid amide
- Carboxamide group
- Carboxylic acid derivative
- Organic nitrogen compound
- Organic oxygen compound
- Organopnictogen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Organonitrogen compound
- Carbonyl group
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0cdr-5974000000-5ab4aa4dfc9d8c43e36a | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00di-9003000000-f0b051d073fd411f6fc8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00di-9010000000-8536375624046cd54fd0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0ab9-9000000000-970ee06935f36f7fdf89 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0019000000-afc917bb7a4f7ea35c85 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-5189000000-bbf718796e416d3ee785 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-006x-9071000000-06eff2193ae0424bbe78 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-3049000000-07d67ca8644a86a3b3f0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-05fr-9011000000-3b58a6d97a98ddfc5dc8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0ab9-9000000000-db65b7eef20488a5ebd4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0019000000-0288513d53364f6bb24a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-4349000000-21a6c9b839d20451a734 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-000f-9161000000-e0da47644f40207bc1b5 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0032032 |
|---|
| FooDB ID | FDB008732 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 8514104 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 10338645 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|