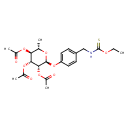| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 23:13:28 UTC |
|---|
| Update Date | 2016-11-09 01:18:20 UTC |
|---|
| Accession Number | CHEM026768 |
|---|
| Identification |
|---|
| Common Name | (Z)-N-[(4-hydroxyphenyl)methyl]ethoxycarbothioamide 4'-(tri-acetylrhamnoside) |
|---|
| Class | Small Molecule |
|---|
| Description | O-Ethyl-4-[(2',3',4'-tri-O-acetyl-alpha-L-rhamnosyloxy)benzyl]thiocarbamate is a fully acetylated thiocarbamate glycoside. It has been isolated from the leaves of Moringa oleifera (horseradish tree). It is found in fats and oils, herbs and spices, and green vegetables. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| O-Ethyl-4-[(2',3',4'-tri-O-acetyl-a-L-rhamnosyloxy)benzyl]thiocarbamate | Generator | | O-Ethyl-4-[(2',3',4'-tri-O-acetyl-a-L-rhamnosyloxy)benzyl]thiocarbamic acid | Generator | | O-Ethyl-4-[(2',3',4'-tri-O-acetyl-alpha-L-rhamnosyloxy)benzyl]thiocarbamic acid | Generator | | O-Ethyl-4-[(2',3',4'-tri-O-acetyl-α-L-rhamnosyloxy)benzyl]thiocarbamate | Generator | | O-Ethyl-4-[(2',3',4'-tri-O-acetyl-α-L-rhamnosyloxy)benzyl]thiocarbamic acid | Generator | | (2S,3R,4R,5S,6S)-4,5-Bis(acetyloxy)-2-[4-({[ethoxy(sulfanyl)methylidene]amino}methyl)phenoxy]-6-methyloxan-3-yl acetic acid | Generator, HMDB | | (2S,3R,4R,5S,6S)-4,5-Bis(acetyloxy)-2-[4-({[ethoxy(sulphanyl)methylidene]amino}methyl)phenoxy]-6-methyloxan-3-yl acetate | Generator, HMDB | | (2S,3R,4R,5S,6S)-4,5-Bis(acetyloxy)-2-[4-({[ethoxy(sulphanyl)methylidene]amino}methyl)phenoxy]-6-methyloxan-3-yl acetic acid | Generator, HMDB | | (Z)-N-[(4-Hydroxyphenyl)methyl]ethoxycarbothioamide 4'-(tri-acetylrhamnoside) | HMDB | | (Z)-N-[(4-Hydroxyphenyl)methyl]ethoxycarbothioamide 4’-(tri-acetylrhamnoside) | HMDB | | (Z)-O-Ethyl 4-[(2',3',4'-tri-O-acetyl-alpha-L-rhamnosyloxy)benzyl]thiocarbamate | HMDB | | (Z)-O-Ethyl 4-[(2’,3’,4’-tri-O-acetyl-α-L-rhamnosyloxy)benzyl]thiocarbamate | HMDB | | (Z)-O-Ethyl-4-[(2',3',4'-tri-O-acetyl-alpha-L-rhamnosyloxy)benzyl]thiocarbamate | HMDB | | (Z)-O-Ethyl-4-[(2’,3’,4’-tri-O-acetyl-α-L-rhamnosyloxy)benzyl]thiocarbamate | HMDB | | N-[(4-Hydroxyphenyl)methyl]ethoxycarbothioamide 4'-(tri-acetylrhamnoside) | HMDB | | N-[(4-Hydroxyphenyl)methyl]ethoxycarbothioamide 4’-(tri-acetylrhamnoside) | HMDB | | O-Ethyl 4-[(2',3',4'-tri-O-acetyl-alpha-L-rhamnosyloxy)benzyl]thiocarbamate | HMDB | | O-Ethyl 4-[(2’,3’,4’-tri-O-acetyl-α-L-rhamnosyloxy)benzyl]thiocarbamate | HMDB | | O-Ethyl-4-[(2',3',4'-tri-O-acetyl-alpha-L-rhamnosyloxy)benzyl]thiocarbamate | HMDB | | O-Ethyl-4-[(2’,3’,4’-tri-O-acetyl-α-L-rhamnosyloxy)benzyl]thiocarbamate | HMDB |
|
|---|
| Chemical Formula | C22H29NO9S |
|---|
| Average Molecular Mass | 483.532 g/mol |
|---|
| Monoisotopic Mass | 483.156 g/mol |
|---|
| CAS Registry Number | 147821-50-9 |
|---|
| IUPAC Name | (2S,3S,4R,5R,6S)-4,5-bis(acetyloxy)-6-(4-{[(ethoxymethanethioyl)amino]methyl}phenoxy)-2-methyloxan-3-yl acetate |
|---|
| Traditional Name | (2S,3S,4R,5R,6S)-4,5-bis(acetyloxy)-6-(4-{[(ethoxymethanethioyl)amino]methyl}phenoxy)-2-methyloxan-3-yl acetate |
|---|
| SMILES | CCO\C(S)=N\CC1=CC=C(O[C@@H]2O[C@@H](C)[C@H](OC(C)=O)[C@@H](OC(C)=O)[C@H]2OC(C)=O)C=C1 |
|---|
| InChI Identifier | InChI=1S/C22H29NO9S/c1-6-27-22(33)23-11-16-7-9-17(10-8-16)32-21-20(31-15(5)26)19(30-14(4)25)18(12(2)28-21)29-13(3)24/h7-10,12,18-21H,6,11H2,1-5H3,(H,23,33)/t12-,18-,19+,20+,21-/m0/s1 |
|---|
| InChI Key | WYFYRQBBNVSDHV-CDEOHPBMSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as phenolic glycosides. These are organic compounds containing a phenolic structure attached to a glycosyl moiety. Some examples of phenolic structures include lignans, and flavonoids. Among the sugar units found in natural glycosides are D-glucose, L-Fructose, and L rhamnose. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbohydrates and carbohydrate conjugates |
|---|
| Direct Parent | Phenolic glycosides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Phenolic glycoside
- Hexose monosaccharide
- O-glycosyl compound
- Tricarboxylic acid or derivatives
- Phenoxy compound
- Phenol ether
- Monocyclic benzene moiety
- Benzenoid
- Monosaccharide
- Oxane
- Thiocarbamic acid ester
- Carboxylic acid ester
- Thiocarbamic acid derivative
- Oxacycle
- Carboxylic acid derivative
- Organoheterocyclic compound
- Acetal
- Organopnictogen compound
- Organic nitrogen compound
- Organic oxide
- Hydrocarbon derivative
- Carbonyl group
- Organonitrogen compound
- Organosulfur compound
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-00di-3221900000-d7626e05bfc544532d9a | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03ec-2364900000-668a7d33526369d69a31 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-097l-0933100000-ffa54f9c26c164c352b3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0cdi-1943000000-1304a42dedd1aa89295f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-9112700000-7f018ed0d9f8b9125b76 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0r00-7439200000-ff0a4e77443d35797cda | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0bt9-9320000000-7af0b411df2249abe32b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-0033900000-d2c5fee3df94ac40062f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-005i-0126900000-930dcedcf095c9b8c0a0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00ec-0955300000-adfc8ca2cc13951fdfd4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-1000900000-2b9b87a4a59d2b15f8cc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-7409400000-a65b544eed6872408423 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0ab9-9606100000-9501a8143adb74ff8f4b | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0031946 |
|---|
| FooDB ID | FDB008637 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 8498489 |
|---|
| ChEBI ID | 172709 |
|---|
| PubChem Compound ID | 10323025 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|