| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 23:08:27 UTC |
|---|
| Update Date | 2016-11-09 01:18:18 UTC |
|---|
| Accession Number | CHEM026642 |
|---|
| Identification |
|---|
| Common Name | Allithiamine |
|---|
| Class | Small Molecule |
|---|
| Description | Allithiamine is found in chives as well as garlic (Allium sativum). It imparts a meaty flavour to foods. Allithiamine has vitamin B1 activity. It has been investigated as a dietary supplement to enhance muscle performance in sports. Allithiamine is a lipid-soluble form of vitamin B1 which occurs naturally in garlic. It is more bioavailable than the water-soluble form of vitamin B1, thiamine, and is the preferred form to be taken in case of a vitamin deficiency.[citation needed |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
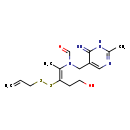
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Allithiamine hydrochloride | MeSH | | N-[(4-amino-2-Methyl-5-pyrimidinyl)methyl]-N-[4-hydroxy-1-methyl-2-(2-propenyldithio)-1-butenyl]formamide, 9ci | HMDB | | TAD | HMDB | | Thiamin-allyl-disulfid | HMDB | | N-[(2E)-5-Hydroxy-3-(prop-2-en-1-yldisulphanyl)pent-2-en-2-yl]-N-[(6-imino-2-methyl-1,6-dihydropyrimidin-5-yl)methyl]formamide | Generator | | Allithiamine | MeSH |
|
|---|
| Chemical Formula | C15H22N4O2S2 |
|---|
| Average Molecular Mass | 354.491 g/mol |
|---|
| Monoisotopic Mass | 354.118 g/mol |
|---|
| CAS Registry Number | 554-44-9 |
|---|
| IUPAC Name | N-[(4-amino-2-methylpyrimidin-5-yl)methyl]-N-[(2E)-5-hydroxy-3-(prop-2-en-1-yldisulfanyl)pent-2-en-2-yl]formamide |
|---|
| Traditional Name | N-[(4-amino-2-methylpyrimidin-5-yl)methyl]-N-[(2E)-5-hydroxy-3-(prop-2-en-1-yldisulfanyl)pent-2-en-2-yl]formamide |
|---|
| SMILES | C\C(N(CC1=C(N)N=C(C)N=C1)C=O)=C(\CCO)SSCC=C |
|---|
| InChI Identifier | InChI=1S/C15H22N4O2S2/c1-4-7-22-23-14(5-6-20)11(2)19(10-21)9-13-8-17-12(3)18-15(13)16/h4,8,10,20H,1,5-7,9H2,2-3H3,(H2,16,17,18)/b14-11+ |
|---|
| InChI Key | WNCAVNGLACHSRZ-SDNWHVSQSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as aminopyrimidines and derivatives. These are organic compounds containing an amino group attached to a pyrimidine ring. Pyrimidine is a 6-membered ring consisting of four carbon atoms and two nitrogen centers at the 1- and 3- ring positions. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Diazines |
|---|
| Sub Class | Pyrimidines and pyrimidine derivatives |
|---|
| Direct Parent | Aminopyrimidines and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Aminopyrimidine
- Imidolactam
- Tertiary carboxylic acid amide
- Heteroaromatic compound
- Amino acid or derivatives
- Carboxamide group
- Organic disulfide
- Allyl sulfur compound
- Azacycle
- Sulfenyl compound
- Carboxylic acid derivative
- Organic nitrogen compound
- Primary amine
- Primary alcohol
- Organosulfur compound
- Organooxygen compound
- Organonitrogen compound
- Alcohol
- Organic oxygen compound
- Amine
- Carbonyl group
- Organopnictogen compound
- Organic oxide
- Hydrocarbon derivative
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-00dl-8933000000-5863edf885acb6aa491d | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-0006-9738100000-988a1868bf948a98f819 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-05g1-4988000000-478fc1fca9712b55c99a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00dl-6955000000-15f9fe0ab0f1565e606e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00di-4900000000-c3dba0a95a3c7199de86 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-1139000000-0d86bae122907dbc2ce7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00di-5090000000-12bed19c4a0f0760784e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9320000000-6f8d2321e8baf30a6d30 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0198000000-93e54a05050e9d040753 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0fy6-1491000000-7f660b8e7968b1da3b39 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00dl-9710000000-21431b57ba2ccc9f0a19 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03dr-1390000000-bd43d3fc6749b4a3a657 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00di-9000000000-cacf3688494c154d1043 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0229-9200000000-8bb76307a0b6cce5fefc | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0031758 |
|---|
| FooDB ID | FDB008429 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00054938 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Allithiamine |
|---|
| Chemspider ID | 14116443 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 12358373 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|