| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 23:06:26 UTC |
|---|
| Update Date | 2016-11-09 01:18:18 UTC |
|---|
| Accession Number | CHEM026593 |
|---|
| Identification |
|---|
| Common Name | 2,4,6-Trimethyl-1,3,5-trithiane |
|---|
| Class | Small Molecule |
|---|
| Description | Modifier for blackcurrant flavou |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
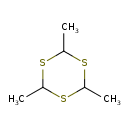
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (2alpha,4alpha,6alpha)-2,4,6-Trimethyl-1,3,5-trithiane | HMDB | | 1,3,5-Trimethyl-2,4,6-trithiane | HMDB | | 1,3,5-Trimethyl-S-trithiane | HMDB | | 1,3,5-Trithiane, 2,4,6-trimethyl, #1 | HMDB | | 1,3,5-Trithiane, 2,4,6-trimethyl, #2 | HMDB | | 2,4,6-Trimethyl-S-trithiane | HMDB | | 2,4,6-Trimethyl-S-trithiane (trithioacetaldehyde) | HMDB | | 2E, 4E,6E-Trimethyl-1,3,5-trithiane | HMDB | | 2E,4E,6E-Trimethyl-1,3,5-trithiane | HMDB | | Thioacetaldehyde | HMDB | | Thioacetaldehyde cyclic trimer | HMDB | | Thioacetaldehyde trimer | HMDB | | Trithioacetaldehyde | HMDB |
|
|---|
| Chemical Formula | C6H12S3 |
|---|
| Average Molecular Mass | 180.354 g/mol |
|---|
| Monoisotopic Mass | 180.010 g/mol |
|---|
| CAS Registry Number | 2765-04-0 |
|---|
| IUPAC Name | 2,4,6-trimethyl-1,3,5-trithiane |
|---|
| Traditional Name | thioacetaldehyde |
|---|
| SMILES | CC1SC(C)SC(C)S1 |
|---|
| InChI Identifier | InChI=1S/C6H12S3/c1-4-7-5(2)9-6(3)8-4/h4-6H,1-3H3 |
|---|
| InChI Key | XQVYLDFSPBXACS-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as trithianes. These are organic compounds containing a six-member aliphatic saturated heterocycle made up of three sulfur atoms and three carbon atoms. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Trithianes |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Trithianes |
|---|
| Alternative Parents | |
|---|
| Substituents | - Trithiane
- Thioacetal
- Dialkylthioether
- Thioether
- Hydrocarbon derivative
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-06v0-9800000000-d31551556b93634e61f4 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-0900000000-fb74d79da7fd047211db | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01p9-9200000000-3f01efed56d1760d2299 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-9100000000-91412bef64559a1a91ea | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0gb9-2900000000-b3af98b622e48cc514aa | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-9100000000-62e5d349c3ea1f73dea5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-9000000000-b566db3687515f5bb73c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-1900000000-6cc262b30f73944f4f68 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-052f-9000000000-2b43af85469b52dd0239 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-9000000000-0f1c40ce2dfc1b8dbc90 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-1900000000-6a3fc6b1d95c2af14965 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0uec-5900000000-dbdf83628211ae483604 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-08fr-9000000000-be75ba92beefd61e0e99 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0031671 |
|---|
| FooDB ID | FDB008331 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 16728 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 17696 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|