| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 22:52:00 UTC |
|---|
| Update Date | 2016-11-09 01:18:14 UTC |
|---|
| Accession Number | CHEM026224 |
|---|
| Identification |
|---|
| Common Name | 6-O-Galloylglucose |
|---|
| Class | Small Molecule |
|---|
| Description | A a galloyl beta-D-glucose compound having a galloyl group at the 6-position. Isolated from the leaves of Sapium sebiferum, it exhibits antihypertensive activity. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
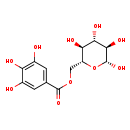
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 6-O-Galloyl-b-D-glucose | Generator | | 6-O-Galloyl-β-D-glucose | Generator |
|
|---|
| Chemical Formula | C13H16O10 |
|---|
| Average Molecular Mass | 332.260 g/mol |
|---|
| Monoisotopic Mass | 332.074 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | [(2R,3S,4S,5R,6R)-3,4,5,6-tetrahydroxyoxan-2-yl]methyl 3,4,5-trihydroxybenzoate |
|---|
| Traditional Name | [(2R,3S,4S,5R,6R)-3,4,5,6-tetrahydroxyoxan-2-yl]methyl 3,4,5-trihydroxybenzoate |
|---|
| SMILES | O[C@@H]1O[C@H](COC(=O)C2=CC(O)=C(O)C(O)=C2)[C@@H](O)[C@H](O)[C@H]1O |
|---|
| InChI Identifier | InChI=1S/C13H16O10/c14-5-1-4(2-6(15)8(5)16)12(20)22-3-7-9(17)10(18)11(19)13(21)23-7/h1-2,7,9-11,13-19,21H,3H2/t7-,9-,10+,11-,13-/m1/s1 |
|---|
| InChI Key | VGVDLJNNDOFWKT-JEUROIALSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as galloyl esters. These are organic compounds that contain an ester derivative of 3,4,5-trihydroxybenzoic acid. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Benzene and substituted derivatives |
|---|
| Sub Class | Benzoic acids and derivatives |
|---|
| Direct Parent | Galloyl esters |
|---|
| Alternative Parents | |
|---|
| Substituents | - Galloyl ester
- P-hydroxybenzoic acid alkyl ester
- M-hydroxybenzoic acid ester
- P-hydroxybenzoic acid ester
- Benzoate ester
- Benzenetriol
- Pyrogallol derivative
- Benzoyl
- 1-hydroxy-4-unsubstituted benzenoid
- 1-hydroxy-2-unsubstituted benzenoid
- Phenol
- Monosaccharide
- Oxane
- Carboxylic acid ester
- Hemiacetal
- Secondary alcohol
- Carboxylic acid derivative
- Oxacycle
- Organoheterocyclic compound
- Monocarboxylic acid or derivatives
- Polyol
- Alcohol
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0hh9-0917000000-6ae6cbbc54c4be152a8b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0w2a-0902000000-f0c05640d102398295e5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0udi-2910000000-74061371339217878d0a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0159-1925000000-af3a2637978d3749d8f8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-014i-1900000000-d59912ac88a4abc52620 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-016r-3900000000-69b70a5ca17ee0655bc4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00l2-0649000000-84c350406562a9b01306 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0w4l-0591000000-5b9f485bc1e1acf9c92a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0ufr-1950000000-30770637042c6bd89bd9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03e9-1596000000-5732446c02c96bbbd220 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00ou-4911000000-a08b60f6ed9346cf45eb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00ou-4910000000-acdea2caa4361eb30141 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0302912 |
|---|
| FooDB ID | FDB006845 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 4476323 |
|---|
| ChEBI ID | 65941 |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|