| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 22:51:36 UTC |
|---|
| Update Date | 2016-11-09 01:18:13 UTC |
|---|
| Accession Number | CHEM026211 |
|---|
| Identification |
|---|
| Common Name | Flavogallol |
|---|
| Class | Small Molecule |
|---|
| Description | Flavogallol is a member of the class of compounds known as hydrolyzable tannins. Hydrolyzable tannins are tannins with a structure characterized by either of the following models. In model 1, the structure contains galloyl units (in some cases, shikimic acid units) are linked to diverse polyol carbohydrate-, catechin-, or triterpenoid units. In model 2, contains at least two galloyl units C-C coupled to each other, and do not contain a glycosidically linked catechin unit. Flavogallol is practically insoluble (in water) and a very weakly acidic compound (based on its pKa). Flavogallol can be found in pomegranate, which makes flavogallol a potential biomarker for the consumption of this food product. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
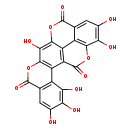
| Chemical Structure | |
|---|
| Synonyms | Not Available |
|---|
| Chemical Formula | C21H8O12 |
|---|
| Average Molecular Mass | 452.281 g/mol |
|---|
| Monoisotopic Mass | 452.002 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | 4,5,6,12,18,19-hexahydroxy-10,14,21-trioxahexacyclo[14.6.2.0²,¹¹.0³,⁸.0¹³,²³.0²⁰,²⁴]tetracosa-1(23),2(11),3(8),4,6,12,16,18,20(24)-nonaene-9,15,22-trione |
|---|
| Traditional Name | 4,5,6,12,18,19-hexahydroxy-10,14,21-trioxahexacyclo[14.6.2.0²,¹¹.0³,⁸.0¹³,²³.0²⁰,²⁴]tetracosa-1(23),2(11),3(8),4,6,12,16,18,20(24)-nonaene-9,15,22-trione |
|---|
| SMILES | OC1=CC2=C(C(O)=C1O)C1=C(OC2=O)C(O)=C2OC(=O)C3=CC(O)=C(O)C4=C3C2=C1C(=O)O4 |
|---|
| InChI Identifier | InChI=1S/C21H8O12/c22-5-1-3-7(14(26)12(5)24)9-11-10-8-4(2-6(23)13(25)16(8)33-21(11)30)20(29)32-18(10)15(27)17(9)31-19(3)28/h1-2,22-27H |
|---|
| InChI Key | JTFOBWXMEHITKZ-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as hydrolyzable tannins. These are tannins with a structure characterized by either of the following models. In model 1, the structure contains galloyl units (in some cases, shikimic acid units) that are linked to diverse polyol carbohydrate-, catechin-, or triterpenoid units. In model 2, contains at least two galloyl units C-C coupled to each other, and do not contain a glycosidically linked catechin unit. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Tannins |
|---|
| Sub Class | Hydrolyzable tannins |
|---|
| Direct Parent | Hydrolyzable tannins |
|---|
| Alternative Parents | |
|---|
| Substituents | - Hydrolyzable tannin
- Ellagic_acid
- Pyranocoumarin
- Linear pyranocoumarin
- Pyranochromene
- 7,8-dihydroxycoumarin
- Coumarin
- Isocoumarin
- Benzopyran
- 1-benzopyran
- 2-benzopyran
- 1-hydroxy-4-unsubstituted benzenoid
- Pyranone
- 1-hydroxy-2-unsubstituted benzenoid
- Pyran
- Benzenoid
- Heteroaromatic compound
- Lactone
- Polyol
- Oxacycle
- Organoheterocyclic compound
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxide
- Organic oxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0000900000-ef4a0e4d3f5d9ddc6388 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0ue9-0000900000-28a6c3012aa9bfdc9fb4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-005c-0009300000-15425e1bba837636ee35 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0zfr-0000900000-0e39cb0f856bece27cec | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0zfr-0001900000-085537164eb4b94d5949 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0ik9-0119500000-628d5f4a8c090604150f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0000900000-5f16f4cde27f8fc21eeb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udi-0000900000-5f16f4cde27f8fc21eeb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-076r-0005900000-f16ea3fb91d267a52114 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0000900000-5ebb1de845c06de1c13c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-0000900000-5c3134f1de84a1bfe4ad | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-03kd-0009200000-d26ad3c391f4272302e2 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0302901 |
|---|
| FooDB ID | FDB006783 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 59696676 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 15559252 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|