| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 22:43:23 UTC |
|---|
| Update Date | 2016-11-09 01:18:11 UTC |
|---|
| Accession Number | CHEM025989 |
|---|
| Identification |
|---|
| Common Name | Ridentin B |
|---|
| Class | Small Molecule |
|---|
| Description | Not Available |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
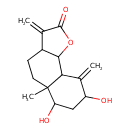
| Chemical Structure | |
|---|
| Synonyms | Not Available |
|---|
| Chemical Formula | C15H20O4 |
|---|
| Average Molecular Mass | 264.317 g/mol |
|---|
| Monoisotopic Mass | 264.136 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | 6,8-dihydroxy-5a-methyl-3,9-dimethylidene-dodecahydronaphtho[1,2-b]furan-2-one |
|---|
| Traditional Name | 6,8-dihydroxy-5a-methyl-3,9-dimethylidene-octahydronaphtho[1,2-b]furan-2-one |
|---|
| SMILES | CC12CCC3C(OC(=O)C3=C)C1C(=C)C(O)CC2O |
|---|
| InChI Identifier | InChI=1S/C15H20O4/c1-7-9-4-5-15(3)11(17)6-10(16)8(2)12(15)13(9)19-14(7)18/h9-13,16-17H,1-2,4-6H2,3H3 |
|---|
| InChI Key | CXFAEUSGWYVIJU-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as eudesmanolides, secoeudesmanolides, and derivatives. These are terpenoids with a structure based on the eudesmanolide (a 3,5a,9-trimethyl-naphtho[1,2-b]furan-2-one derivative) or secoeudesmanolide (a 3,6-dimethyl-5-(pentan-2-yl)-1-benzofuran-2-one derivative) skeleton. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Terpene lactones |
|---|
| Direct Parent | Eudesmanolides, secoeudesmanolides, and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Eudesmanolide
- Sesquiterpenoid
- Naphthofuran
- Gamma butyrolactone
- Cyclic alcohol
- Tetrahydrofuran
- Enoate ester
- Alpha,beta-unsaturated carboxylic ester
- Secondary alcohol
- Lactone
- Carboxylic acid ester
- Oxacycle
- Monocarboxylic acid or derivatives
- Carboxylic acid derivative
- Organoheterocyclic compound
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxide
- Organic oxygen compound
- Alcohol
- Carbonyl group
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00kb-0190000000-6c1af35bfacb7e6cc985 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00mk-0950000000-36648678f1e86eccde15 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0gdu-5910000000-904abc979d0ffaf1f15c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0090000000-4c5abe6976fb689b2107 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0292-0090000000-41940ff3842355d70acd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0i01-3950000000-5677afbb8961ec4925b8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00kb-0190000000-c3ae3c936234ffafb2d0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00kb-0690000000-650f682abc4cc73ef893 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-002u-1920000000-0be46a019dee8bd22c4d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0090000000-396ced35e5711708c4bf | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03xu-2390000000-cf0d41affe8cfb782492 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4j-7590000000-149934c6d8e66fb75b90 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0302707 |
|---|
| FooDB ID | FDB005895 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 28565358 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 14779601 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|