| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 22:41:53 UTC |
|---|
| Update Date | 2016-11-09 01:18:11 UTC |
|---|
| Accession Number | CHEM025951 |
|---|
| Identification |
|---|
| Common Name | Ciliaric acid |
|---|
| Class | Small Molecule |
|---|
| Description | Ciliaric acid, also known as ciliarate, is a member of the class of compounds known as kaurane diterpenoids. Kaurane diterpenoids are diterpene alkaloids with a structure that is based on the kaurane skeleton. Kaurane is a tetracyclic compound that arises by cyclisation of a pimarane precursor followed by rearrangement. It possesses a [3,2,1]-bicyclic ring system with C15-C16 bridge connected to C13, forming the five-membered ring D. Ciliaric acid is practically insoluble (in water) and a weakly acidic compound (based on its pKa). Ciliaric acid can be found in sunflower, which makes ciliaric acid a potential biomarker for the consumption of this food product. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
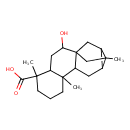
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Ciliarate | Generator |
|
|---|
| Chemical Formula | C20H30O3 |
|---|
| Average Molecular Mass | 318.450 g/mol |
|---|
| Monoisotopic Mass | 318.219 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | 2-hydroxy-5,9,13-trimethylpentacyclo[11.2.1.0¹,¹⁰.0⁴,⁹.0¹²,¹⁴]hexadecane-5-carboxylic acid |
|---|
| Traditional Name | 2-hydroxy-5,9,13-trimethylpentacyclo[11.2.1.0¹,¹⁰.0⁴,⁹.0¹²,¹⁴]hexadecane-5-carboxylic acid |
|---|
| SMILES | CC12CC34CC1C2CC3C1(C)CCCC(C)(C1CC4O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C20H30O3/c1-17-5-4-6-18(2,16(22)23)13(17)8-15(21)20-9-12-11(7-14(17)20)19(12,3)10-20/h11-15,21H,4-10H2,1-3H3,(H,22,23) |
|---|
| InChI Key | REFPIPGRFRVTDA-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as kaurane diterpenoids. These are diterpene alkaloids with a structure that is based on the kaurane skeleton. Kaurane is a tetracyclic compound that arises by cyclisation of a pimarane precursor followed by rearrangement. It possesses a [3,2,1]-bicyclic ring system with C15-C16 bridge connected to C13, forming the five-membered ring D. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Diterpenoids |
|---|
| Direct Parent | Kaurane diterpenoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Atisane diterpenoid
- Kaurane diterpenoid
- Villanovane, atisane, trachylobane or helvifulvane diterpenoid
- Alkaloid or derivatives
- Cyclic alcohol
- Secondary alcohol
- Carboxylic acid derivative
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Organic oxide
- Alcohol
- Carbonyl group
- Organic oxygen compound
- Organooxygen compound
- Hydrocarbon derivative
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0uxr-0049000000-520df8d942fa56e09a73 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-106r-0294000000-3b3d68e9475e3eed0db5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-014i-3960000000-88553521233561dbea4c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-0069000000-08ed6130104893d75b4c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-06di-0093000000-5a1e50fdb6cb8164720a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-1091000000-1036c3395ebd7219baa7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-01b9-0049000000-c70b4ec8b8c1449834b8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00xr-0983000000-4c3f1f37632debbcc6e6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0avi-3920000000-3251aecab449020e43a1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-0009000000-062ad2a35e11d303cf23 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-014i-0009000000-062ad2a35e11d303cf23 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-014i-1009000000-7e11fc3c22d4c3fa8f87 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0302679 |
|---|
| FooDB ID | FDB005712 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 73028428 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|