| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 22:40:31 UTC |
|---|
| Update Date | 2016-11-09 01:18:10 UTC |
|---|
| Accession Number | CHEM025909 |
|---|
| Identification |
|---|
| Common Name | Quercetin 3-O-beta-D-2-glucosyl-rutinoside |
|---|
| Class | Small Molecule |
|---|
| Description | Not Available |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
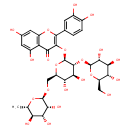
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3',4',5,7-Tetrahydroxy-3-[(2-O-b-D-glucopyranosyl-6-O-a-L-rhamnopyranosyl-b-D-glucopyranosyl)oxy]flavone | Generator | | 3',4',5,7-Tetrahydroxy-3-[(2-O-β-D-glucopyranosyl-6-O-α-L-rhamnopyranosyl-β-D-glucopyranosyl)oxy]flavone | Generator | | Quercetin 3-O-b-D-2-glucosyl-rutinoside | Generator | | Quercetin 3-O-β-D-2-glucosyl-rutinoside | Generator |
|
|---|
| Chemical Formula | C33H40O21 |
|---|
| Average Molecular Mass | 772.658 g/mol |
|---|
| Monoisotopic Mass | 772.206 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | 3-{[(2S,3R,4S,5S,6R)-4,5-dihydroxy-3-{[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}-6-({[(2R,3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxy}methyl)oxan-2-yl]oxy}-2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-4H-chromen-4-one |
|---|
| Traditional Name | 3-{[(2S,3R,4S,5S,6R)-4,5-dihydroxy-3-{[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}-6-({[(2R,3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxy}methyl)oxan-2-yl]oxy}-2-(3,4-dihydroxyphenyl)-5,7-dihydroxychromen-4-one |
|---|
| SMILES | C[C@@H]1O[C@@H](OC[C@H]2O[C@@H](OC3=C(OC4=C(C(O)=CC(O)=C4)C3=O)C3=CC(O)=C(O)C=C3)[C@H](O[C@@H]3O[C@H](CO)[C@@H](O)[C@H](O)[C@H]3O)[C@@H](O)[C@@H]2O)[C@H](O)[C@H](O)[C@H]1O |
|---|
| InChI Identifier | InChI=1S/C33H40O21/c1-9-19(39)23(43)26(46)31(49-9)48-8-17-21(41)25(45)30(54-32-27(47)24(44)20(40)16(7-34)51-32)33(52-17)53-29-22(42)18-14(38)5-11(35)6-15(18)50-28(29)10-2-3-12(36)13(37)4-10/h2-6,9,16-17,19-21,23-27,30-41,43-47H,7-8H2,1H3/t9-,16+,17+,19-,20+,21+,23+,24-,25-,26+,27+,30+,31+,32-,33-/m0/s1 |
|---|
| InChI Key | MNMUPTOJETVJCW-WHBTYZLHSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as rotenones. These are rotenoids with a structure based on a 6a,12a-dihydrochromeno[3,4-b]chromen-12(6H)-one skeleton. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Isoflavonoids |
|---|
| Sub Class | Rotenoids |
|---|
| Direct Parent | Rotenones |
|---|
| Alternative Parents | |
|---|
| Substituents | - Rotenone or derivatives
- 8-prenylated isoflavanone
- Isoflavanone
- Isoflavan
- Pyranochromene
- 2,2-dimethyl-1-benzopyran
- Chromone
- Chromane
- Benzopyran
- 1-benzopyran
- Anisole
- Aryl alkyl ketone
- Aryl ketone
- Alkyl aryl ether
- Benzenoid
- Ketone
- Organoheterocyclic compound
- Oxacycle
- Ether
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxide
- Organic oxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0uk9-4917254700-7031888d05396a21ecb6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-3927011100-4fad5160b3e1eb699613 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0ufr-4926000000-8a2f5f1ce653c2436560 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-0000000900-ec3d348581783841d73a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0fk9-0005000900-06a06e12d9d999109ccf | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0udi-0019000100-6b0b22c02d3a0356199c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-114l-0339144700-1383e4e3442d0c3ad960 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udi-0239322000-1303cee17cccf82c63ea | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0zg0-1958211200-3648f97384566d16b242 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0009000200-0beb8c5f0064cf6981c5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0ur0-0009000900-97e56086872ef552eae3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0udi-0009000000-58b2754f892c40f07699 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0302651 |
|---|
| FooDB ID | FDB005614 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 59696371 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 100913778 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|