| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 22:39:47 UTC |
|---|
| Update Date | 2016-11-09 01:18:10 UTC |
|---|
| Accession Number | CHEM025887 |
|---|
| Identification |
|---|
| Common Name | 6-(4-O-beta-D-Glucosyl-3-methyl-trans-but-2-enyl-amino)-purine |
|---|
| Class | Small Molecule |
|---|
| Description | The trans-isomer of O-beta-D-glucosylzeatin. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
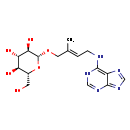
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| O-beta-D-Glucosylzeatin | ChEBI | | trans-Zeatin-O-glucoside | ChEBI | | O-b-D-Glucosylzeatin | Generator | | O-Β-D-glucosylzeatin | Generator | | O-b-D-Glucosyl-trans-zeatin | Generator | | O-Β-D-glucosyl-trans-zeatin | Generator | | 6-(4-O-b-D-Glucosyl-3-methyl-trans-but-2-enyl-amino)-purine | Generator | | 6-(4-O-β-D-glucosyl-3-methyl-trans-but-2-enyl-amino)-purine | Generator | | O-beta-D-Glucosyl-trans-zeatin | KEGG |
|
|---|
| Chemical Formula | C16H23N5O6 |
|---|
| Average Molecular Mass | 381.384 g/mol |
|---|
| Monoisotopic Mass | 381.165 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | (2R,3S,4S,5R,6R)-2-(hydroxymethyl)-6-{[(2E)-2-methyl-4-[(1H-purin-6-yl)amino]but-2-en-1-yl]oxy}oxane-3,4,5-triol |
|---|
| Traditional Name | (2R,3S,4S,5R,6R)-2-(hydroxymethyl)-6-{[(2E)-2-methyl-4-(1H-purin-6-ylamino)but-2-en-1-yl]oxy}oxane-3,4,5-triol |
|---|
| SMILES | C\C(CO[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O)=C/CNC1=NC=NC2=C1NC=N2 |
|---|
| InChI Identifier | InChI=1S/C16H23N5O6/c1-8(2-3-17-14-10-15(19-6-18-10)21-7-20-14)5-26-16-13(25)12(24)11(23)9(4-22)27-16/h2,6-7,9,11-13,16,22-25H,3-5H2,1H3,(H2,17,18,19,20,21)/b8-2+/t9-,11-,12+,13-,16-/m1/s1 |
|---|
| InChI Key | UUPDCCPAOMDMPT-HNVSNYHQSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as fatty acyl glycosides of mono- and disaccharides. Fatty acyl glycosides of mono- and disaccharides are compounds composed of a mono- or disaccharide moiety linked to one hydroxyl group of a fatty alcohol or of a phosphorylated alcohol (phosphoprenols), a hydroxy fatty acid or to one carboxyl group of a fatty acid (ester linkage) or to an amino alcohol. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Fatty Acyls |
|---|
| Sub Class | Fatty acyl glycosides |
|---|
| Direct Parent | Fatty acyl glycosides of mono- and disaccharides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Fatty acyl glycoside of mono- or disaccharide
- Alkyl glycoside
- Glycosyl compound
- O-glycosyl compound
- 6-alkylaminopurine
- 6-aminopurine
- Imidazopyrimidine
- Purine
- Aminopyrimidine
- Secondary aliphatic/aromatic amine
- Monosaccharide
- Oxane
- Pyrimidine
- Imidolactam
- Azole
- Heteroaromatic compound
- Imidazole
- Secondary alcohol
- Organoheterocyclic compound
- Secondary amine
- Polyol
- Acetal
- Azacycle
- Oxacycle
- Primary alcohol
- Organic nitrogen compound
- Hydrocarbon derivative
- Organonitrogen compound
- Organopnictogen compound
- Amine
- Alcohol
- Organooxygen compound
- Organic oxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0w30-0298000000-678c0fd32c5a316fdde2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udr-4982000000-93e6fbf8b77b7ed05948 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0f79-9730000000-769ec979a442986534a3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-001i-1439000000-01b1d4b9c22163da5e55 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-01q9-3924000000-ca0daf6896587aa129aa | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-053u-8910000000-a06ad60ece93e54d294d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-0019000000-2c70425e036b227bbae3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udi-0292000000-47b6e456feb6c07e2b11 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0f79-3890000000-cef5c68fcd71b09170ee | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-001i-0319000000-09a2cada4b696df94078 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0j4i-5389000000-7ffde7d78e85e3603b5f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-053r-5910000000-86999dda86df00397445 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0302640 |
|---|
| FooDB ID | FDB005542 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 4574478 |
|---|
| ChEBI ID | 38266 |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | C03423 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|