| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 22:31:54 UTC |
|---|
| Update Date | 2016-11-09 01:18:08 UTC |
|---|
| Accession Number | CHEM025660 |
|---|
| Identification |
|---|
| Common Name | 5-Hydroxycapsanthin-3,6-epoxide |
|---|
| Class | Small Molecule |
|---|
| Description | 5-hydroxycapsanthin-3,6-epoxide is a member of the class of compounds known as xanthophylls. Xanthophylls are carotenoids containing an oxygenated carotene backbone. Carotenes are characterized by the presence of two end-groups (mostly cyclohexene rings, but also cyclopentene rings or acyclic groups) linked by a long branched alkyl chain. Carotenes belonging form a subgroup of the carotenoids family. Xanthophylls arise by oxygenation of the carotene backbone. Thus, 5-hydroxycapsanthin-3,6-epoxide is considered to be an isoprenoid lipid molecule. 5-hydroxycapsanthin-3,6-epoxide is practically insoluble (in water) and a very weakly acidic compound (based on its pKa). 5-hydroxycapsanthin-3,6-epoxide can be found in a number of food items such as yellow bell pepper, pepper (c. annuum), green bell pepper, and orange bell pepper, which makes 5-hydroxycapsanthin-3,6-epoxide a potential biomarker for the consumption of these food products. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
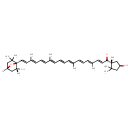
| Chemical Structure | |
|---|
| Synonyms | Not Available |
|---|
| Chemical Formula | C40H56O4 |
|---|
| Average Molecular Mass | 600.870 g/mol |
|---|
| Monoisotopic Mass | 600.418 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | (2E,4E,6E,8E,10E,12E,14E,16E,18E)-1-[(1R,4S)-4-hydroxy-1,2,2-trimethylcyclopentyl]-19-[(1S,2R,4R)-2-hydroxy-2,6,6-trimethyl-7-oxabicyclo[2.2.1]heptan-1-yl]-4,8,13,17-tetramethylnonadeca-2,4,6,8,10,12,14,16,18-nonaen-1-one |
|---|
| Traditional Name | (2E,4E,6E,8E,10E,12E,14E,16E,18E)-1-[(1R,4S)-4-hydroxy-1,2,2-trimethylcyclopentyl]-19-[(1S,2R,4R)-2-hydroxy-2,6,6-trimethyl-7-oxabicyclo[2.2.1]heptan-1-yl]-4,8,13,17-tetramethylnonadeca-2,4,6,8,10,12,14,16,18-nonaen-1-one |
|---|
| SMILES | O[C@H]1CC(C)(C)[C@@](C)(C1)C(=O)\C=C\C(\C)=C\C=C\C(\C)=C\C=C\C=C(/C)\C=C\C=C(/C)\C=C\[C@]12O[C@]([H])(CC1(C)C)C[C@@]2(C)O |
|---|
| InChI Identifier | InChI=1S/C40H56O4/c1-29(17-13-19-31(3)21-22-35(42)38(9)26-33(41)25-36(38,5)6)15-11-12-16-30(2)18-14-20-32(4)23-24-40-37(7,8)27-34(44-40)28-39(40,10)43/h11-24,33-34,41,43H,25-28H2,1-10H3/b12-11+,17-13+,18-14+,22-21+,24-23+,29-15+,30-16+,31-19+,32-20+/t33-,34-,38-,39+,40+/m0/s1 |
|---|
| InChI Key | TWTPPPZIWNGQCQ-YNKJSWFTSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as xanthophylls. These are carotenoids containing an oxygenated carotene backbone. Carotenes are characterized by the presence of two end-groups (mostly cyclohexene rings, but also cyclopentene rings or acyclic groups) linked by a long branched alkyl chain. Carotenes belonging form a subgroup of the carotenoids family. Xanthophylls arise by oxygenation of the carotene backbone. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Tetraterpenoids |
|---|
| Direct Parent | Xanthophylls |
|---|
| Alternative Parents | |
|---|
| Substituents | - Xanthophyll
- Cyclopentanol
- Monosaccharide
- Acryloyl-group
- Cyclic alcohol
- Enone
- Tertiary alcohol
- Tetrahydrofuran
- Alpha,beta-unsaturated ketone
- Secondary alcohol
- Ketone
- Ether
- Dialkyl ether
- Organoheterocyclic compound
- Oxacycle
- Organooxygen compound
- Organic oxide
- Carbonyl group
- Alcohol
- Hydrocarbon derivative
- Organic oxygen compound
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-0251794000-8afb77f5dd86d9100711 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-07cr-0493530000-991cad0eea1eb8e5f13e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-06vi-2984300000-c2cafaa7bdd80efdc4d9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-0100190000-9470f729c67ea1992904 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-000t-0400390000-fdda1b8fb11f8f97812d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0k9i-1900260000-e55f6102d3ff02a59867 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | FDB004638 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 11734696 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|