| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 22:19:12 UTC |
|---|
| Update Date | 2016-11-09 01:18:03 UTC |
|---|
| Accession Number | CHEM025319 |
|---|
| Identification |
|---|
| Common Name | cis-Ajoene |
|---|
| Class | Small Molecule |
|---|
| Description | Ajoene is found in onion-family vegetables. Ajoene is isolated from garlic (Allium sativum) extracts. Nutriceutical with anti-cancer properties Ajoene is a chemical compound available from garlic (Allium sativum). The name (and pronunciation) is derived from "ajo", the Spanish word for garlic. It is found as a mixture of two isomers, E-, and Z- 4,5,9-trithiadodeca-1,6,11-triene 9-oxide. Ajoene, an unsaturated disulfide, is formed from the bonding of three allicin molecules. Allicin is a sulfinyl compound that gives garlic its strong odor and flavor. The release of allicin occurs after a garlic clove is crushed or finely chopped. Subsequent formation of ajoene occurs when allicin is dissolved in various solvents including edible oils. Ajoene is also found in garlic extract. Ajoene is most stable and most abundant in macerate of garlic (chopped garlic in edible oil) |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
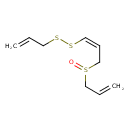
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 2-Propenyl 3-(2-propenylsulfinyl)-1-propenyl disulfide, 9ci | HMDB | | 2-Propenyl-3-(2-propenylsulfinyl)-1-propenyl disulfide | HMDB | | 4,5,9-Trithia-1,6,11-dodecatriene 9-oxide | HMDB | | 4,5,9-Trithiadodeca-1,6,11-triene 9-oxide | HMDB | | Allyl 3-allylsulfinyl-1-propenyl disulfide | HMDB | | Disulfide, 2-propenyl 3-(2-propenylsulfinyl)-1-propenyl | HMDB | | trans-Ajoene | HMDB |
|
|---|
| Chemical Formula | C9H14OS3 |
|---|
| Average Molecular Mass | 234.402 g/mol |
|---|
| Monoisotopic Mass | 234.021 g/mol |
|---|
| CAS Registry Number | 92285-01-3 |
|---|
| IUPAC Name | 3-{[(1Z)-3-(prop-2-ene-1-sulfinyl)prop-1-en-1-yl]disulfanyl}prop-1-ene |
|---|
| Traditional Name | 3-{[(1Z)-3-(prop-2-ene-1-sulfinyl)prop-1-en-1-yl]disulfanyl}prop-1-ene |
|---|
| SMILES | C=CCSS\C=C/CS(=O)CC=C |
|---|
| InChI Identifier | InChI=1S/C9H14OS3/c1-3-6-11-12-7-5-9-13(10)8-4-2/h3-5,7H,1-2,6,8-9H2/b7-5- |
|---|
| InChI Key | IXELFRRANAOWSF-ALCCZGGFSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as sulfoxides. Sulfoxides are compounds containing a sulfoxide functional group, with the structure RS(=O)R' (R,R' not H). |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organosulfur compounds |
|---|
| Class | Sulfoxides |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Sulfoxides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Allyl sulfur compound
- Sulfoxide
- Organic disulfide
- Sulfenyl compound
- Sulfinyl compound
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0006-9300000000-38a71ef9ab244c72057d | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000l-7890000000-11928f7c30159af165e8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0006-9600000000-c607fc1496412765e8c5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-006x-9200000000-a2836ecdf1469af5047c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0016-6960000000-e17a4526d3baecc4e388 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0079-9300000000-d62719e8491d22e01b5c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0f72-9300000000-e0cce5cae13d413bf294 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000i-5940000000-2ff7b7a62d176c1fb383 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0079-9400000000-cb9a19f42f5eeb7c8908 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00di-9000000000-c468260d51fc7a1b3b0d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-9310000000-a37b89564227f0a2bab0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00dr-9200000000-110a3f385d2a123a74ca | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-000i-9100000000-1d553532a99796a2a70b | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0033566 |
|---|
| FooDB ID | FDB003580 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00052510 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Ajoene |
|---|
| Chemspider ID | 8056824 |
|---|
| ChEBI ID | 80707 |
|---|
| PubChem Compound ID | 9881148 |
|---|
| Kegg Compound ID | C16757 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | | 1. Naganawa R, Iwata N, Ishikawa K, Fukuda H, Fujino T, Suzuki A: Inhibition of microbial growth by ajoene, a sulfur-containing compound derived from garlic. Appl Environ Microbiol. 1996 Nov;62(11):4238-42. | | 2. Ledezma E, Apitz-Castro R: [Ajoene the main active compound of garlic (Allium sativum): a new antifungal agent]. Rev Iberoam Micol. 2006 Jun;23(2):75-80. | | 3. Urbina JA, Marchan E, Lazardi K, Visbal G, Apitz-Castro R, Gil F, Aguirre T, Piras MM, Piras R: Inhibition of phosphatidylcholine biosynthesis and cell proliferation in Trypanosoma cruzi by ajoene, an antiplatelet compound isolated from garlic. Biochem Pharmacol. 1993 Jun 22;45(12):2381-7. | | 4. Kaschula CH, Hunter R, Parker MI: Garlic-derived anticancer agents: structure and biological activity of ajoene. Biofactors. 2010 Jan-Feb;36(1):78-85. doi: 10.1002/biof.76. | | 5. Srivastava KC, Tyagi OD: Effects of a garlic-derived principle (ajoene) on aggregation and arachidonic acid metabolism in human blood platelets. Prostaglandins Leukot Essent Fatty Acids. 1993 Aug;49(2):587-95. | | 6. Yannai, Shmuel. (2004) Dictionary of food compounds with CD-ROM: Additives, flavors, and ingredients. Boca Raton: Chapman & Hall/CRC. |
|
|---|