| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 22:15:16 UTC |
|---|
| Update Date | 2016-11-09 01:18:02 UTC |
|---|
| Accession Number | CHEM025237 |
|---|
| Identification |
|---|
| Common Name | Oleoside dimethyl ester |
|---|
| Class | Small Molecule |
|---|
| Description | Oleoside dimethyl ester is found in fats and oils. Oleoside dimethyl ester is a constituent of Olea europaea (olive) |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
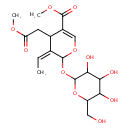
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (-)-Oleoside dimethyl ester | HMDB | | Secologanoside 7-methyl ester | HMDB | | Methyl 3-ethylidene-4-(2-methoxy-2-oxoethyl)-2-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}-3,4-dihydro-2H-pyran-5-carboxylic acid | Generator |
|
|---|
| Chemical Formula | C18H26O11 |
|---|
| Average Molecular Mass | 418.392 g/mol |
|---|
| Monoisotopic Mass | 418.148 g/mol |
|---|
| CAS Registry Number | 30164-95-5 |
|---|
| IUPAC Name | methyl (3Z)-3-ethylidene-4-(2-methoxy-2-oxoethyl)-2-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}-3,4-dihydro-2H-pyran-5-carboxylate |
|---|
| Traditional Name | methyl (5Z)-5-ethylidene-4-(2-methoxy-2-oxoethyl)-6-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}-4,6-dihydropyran-3-carboxylate |
|---|
| SMILES | COC(=O)CC1\C(=C\C)C(OC2OC(CO)C(O)C(O)C2O)OC=C1C(=O)OC |
|---|
| InChI Identifier | InChI=1S/C18H26O11/c1-4-8-9(5-12(20)25-2)10(16(24)26-3)7-27-17(8)29-18-15(23)14(22)13(21)11(6-19)28-18/h4,7,9,11,13-15,17-19,21-23H,5-6H2,1-3H3/b8-4- |
|---|
| InChI Key | KYVUMEGNMQDSHO-YWEYNIOJSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as terpene glycosides. These are prenol lipids containing a carbohydrate moiety glycosidically bound to a terpene backbone. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Terpene glycosides |
|---|
| Direct Parent | Terpene glycosides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Terpene glycoside
- Glycosyl compound
- Secoiridoid-skeleton
- O-glycosyl compound
- Monocyclic monoterpenoid
- Monoterpenoid
- Sugar acid
- Dicarboxylic acid or derivatives
- Monosaccharide
- Oxane
- Vinylogous ester
- Enoate ester
- Methyl ester
- Alpha,beta-unsaturated carboxylic ester
- Secondary alcohol
- Carboxylic acid ester
- Polyol
- Organoheterocyclic compound
- Oxacycle
- Carboxylic acid derivative
- Acetal
- Primary alcohol
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Alcohol
- Carbonyl group
- Organooxygen compound
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0kg9-6409200000-11262ee5471390675baa | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-01b9-2583059000-8e6b42abbe303bbf36e9 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-07e0-0973300000-bb39fd472b5fe839723a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0bvr-0970000000-9bac481efae3abe54d22 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-002k-4910000000-bcf786108b1a8bb4f119 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-066r-1394400000-092f7553eb76dffcac0f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-074i-2493000000-22ed019237cec8cea682 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-074l-5950000000-106277dbe29ec2075190 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-0595700000-b4e6e8a828a72063d900 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00or-1739100000-ad216f04d4472989ba2e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-000w-2920000000-d49403b323fe52c4a0c6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-052b-0970100000-7a8f3d442efadecc600c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-069s-0930000000-f1ece553b991001d9e55 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0002-4902000000-89359b8aaabebcf37f05 | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0031350 |
|---|
| FooDB ID | FDB003414 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00033266 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 14038297 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|