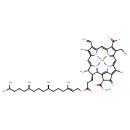
Identification Common Name Chlorophyll b Class Small Molecule Description Contaminant Sources Contaminant Type Not Available Chemical Structure Synonyms Value Source (SP-4-2)-((2E,7R,11R)-3,7,11,15-Tetramethyl-2-hexadecenyl (3S,4S,21R)-9-ethenyl-14-ethyl-13-formyl-21-(methoxycarbonyl)-4,8,18-trimethyl-20-oxo-3-phorbinepropanoato(2-)-kappan(23),kappan(24),kappan(25),kappan(26))-magnesium ChEBI Chlorophyll b MeSH
Chemical Formula C55 H71 MgN4 O6 Average Molecular Mass 908.499 g/mol Monoisotopic Mass 907.522 g/mol CAS Registry Number 519-62-0 IUPAC Name (5R,22S,23S)-17-ethenyl-12-ethyl-13-formyl-5-(methoxycarbonyl)-8,18,22-trimethyl-6-oxo-23-(3-oxo-3-{[(2E,7R,11R)-3,7,11,15-tetramethylhexadec-2-en-1-yl]oxy}propyl)-2,25lambda5,26lambda5,27-tetraaza-1-magnesanonacyclo[12.11.1.1^{1,16}.0^{2,9}.0^{3,7}.0^{4,24}.0^{11,26}.0^{21,25}.0^{19,27}]heptacosa-3,7,9,11(26),12,14,16,18,20,24-decaene-25,26-bis(ylium)-1,1-diuide Traditional Name (5R,22S,23S)-17-ethenyl-12-ethyl-13-formyl-5-(methoxycarbonyl)-8,18,22-trimethyl-6-oxo-23-(3-oxo-3-{[(2E,7R,11R)-3,7,11,15-tetramethylhexadec-2-en-1-yl]oxy}propyl)-2,25lambda5,26lambda5,27-tetraaza-1-magnesanonacyclo[12.11.1.1^{1,16}.0^{2,9}.0^{3,7}.0^{4,24}.0^{11,26}.0^{21,25}.0^{19,27}]heptacosa-3,7,9,11(26),12,14,16,18,20,24-decaene-25,26-bis(ylium)-1,1-diuide SMILES [Mg++].[H]\C(COC(=O)CCC1C(C)C2=N\C1=C1/C(C(=O)OC)C(=O)C3=C(C)C(=CC4=C(CC)C(=CO)C(C=C5[N-]C(=C2)C(C)=C5C=C)=N4)N=C13)=C(\C)CCCC(C)CCCC(C)CCCC(C)C InChI Identifier InChI=1S/C55H72N4O6.Mg/c1-12-38-35(8)42-27-43-36(9)40(23-24-48(61)65-26-25-34(7)22-16-21-33(6)20-15-19-32(5)18-14-17-31(3)4)52(58-43)50-51(55(63)64-11)54(62)49-37(10)44(59-53(49)50)28-46-39(13-2)41(30-60)47(57-46)29-45(38)56-42;/h12,25,27-33,36,40,51H,1,13-24,26H2,2-11H3,(H2,56,57,58,59,60,62);/q;+2/p-1/b34-25+; InChI Key MSLKMRUEVOYOOZ-HXAPHQIMSA-M