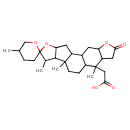| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 22:01:17 UTC |
|---|
| Update Date | 2016-11-09 01:17:59 UTC |
|---|
| Accession Number | CHEM024931 |
|---|
| Identification |
|---|
| Common Name | 2,3-Secoporrigenin |
|---|
| Class | Small Molecule |
|---|
| Description | Constituent of Allium porrum (leek). (6beta,25S)-2,3-Secospirostan-3,6-olid-2-oic acid is found in onion-family vegetables. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 2-{5,7',9',13'-tetramethyl-16'-oxo-5',17'-dioxaspiro[oxane-2,6'-pentacyclo[10.7.0.0²,⁹.0⁴,⁸.0¹⁴,¹⁸]nonadecane]-13'-yl}acetate | Generator |
|
|---|
| Chemical Formula | C27H40O6 |
|---|
| Average Molecular Mass | 460.603 g/mol |
|---|
| Monoisotopic Mass | 460.282 g/mol |
|---|
| CAS Registry Number | 189014-46-8 |
|---|
| IUPAC Name | 2-{5,7',9',13'-tetramethyl-16'-oxo-5',17'-dioxaspiro[oxane-2,6'-pentacyclo[10.7.0.0²,⁹.0⁴,⁸.0¹⁴,¹⁸]nonadecane]-13'-yl}acetic acid |
|---|
| Traditional Name | 5,7',9',13'-tetramethyl-16'-oxo-5',17'-dioxaspiro[oxane-2,6'-pentacyclo[10.7.0.0²,⁹.0⁴,⁸.0¹⁴,¹⁸]nonadecane]-13'-ylacetic acid |
|---|
| SMILES | CC1C2C(CC3C4CC5OC(=O)CC5C(C)(CC(O)=O)C4CCC23C)OC11CCC(C)CO1 |
|---|
| InChI Identifier | InChI=1S/C27H40O6/c1-14-5-8-27(31-13-14)15(2)24-21(33-27)10-18-16-9-20-19(11-23(30)32-20)26(4,12-22(28)29)17(16)6-7-25(18,24)3/h14-21,24H,5-13H2,1-4H3,(H,28,29) |
|---|
| InChI Key | OTCSBULKTTUVHL-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as naphthofurans. Naphthofurans are compounds containing a furan ring fused to a naphthalene moiety. Furan is a 5 membered- ring aromatic ring with four carbon and one oxygen atoms. Naphthalene is a polycyclic aromatic hydrocarbon made up of two fused benzene rings. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Naphthofurans |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Naphthofurans |
|---|
| Alternative Parents | Not Available |
|---|
| Substituents | Not Available |
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-016r-4085900000-3ed2af08dc2e14c14a72 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-014i-6133950000-071806b2b22010b0e18e | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-02tc-3003900000-891cd1aadd1d390bc95c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01b9-6097600000-de1f403c3b807ac4064f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-014i-9023000000-1cb4a1c205808131c2f0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-066r-3002900000-6ce014db014e4636aaa7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-066r-3008900000-71694992a6561b28d11a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-014l-9004100000-79ca61bd15d93aac1d44 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0000900000-ab4c1560f15ff18d432e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-2000900000-056b6805a872ac237c47 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4l-3002900000-9ca7c161c7c6ca20d456 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03dl-0000900000-b8224740cdb5bda73a6a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0j4i-0014900000-6d212a4625881f7d9f34 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0fr2-4359300000-1c9916ab8f4e9f6f786b | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0030909 |
|---|
| FooDB ID | FDB002875 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 78385463 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|