| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 22:00:50 UTC |
|---|
| Update Date | 2016-11-09 01:17:58 UTC |
|---|
| Accession Number | CHEM024923 |
|---|
| Identification |
|---|
| Common Name | Eremopetasitenin A1 |
|---|
| Class | Small Molecule |
|---|
| Description | Eremopetasitenin B1 is found in green vegetables. Eremopetasitenin B1 is a constituent of Petasites japonicus (sweet coltsfoot) |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
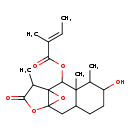
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 5-Hydroxy-3,4,13-trimethyl-12-oxo-11,14-dioxatetracyclo[8.3.1.0¹,¹⁰.0³,⁸]tetradecan-2-yl (2E)-2-methylbut-2-enoic acid | HMDB |
|
|---|
| Chemical Formula | C20H28O6 |
|---|
| Average Molecular Mass | 364.433 g/mol |
|---|
| Monoisotopic Mass | 364.189 g/mol |
|---|
| CAS Registry Number | 71047-01-3 |
|---|
| IUPAC Name | 5-hydroxy-3,4,13-trimethyl-12-oxo-11,14-dioxatetracyclo[8.3.1.0¹,¹⁰.0³,⁸]tetradecan-2-yl (2E)-2-methylbut-2-enoate |
|---|
| Traditional Name | 5-hydroxy-3,4,13-trimethyl-12-oxo-11,14-dioxatetracyclo[8.3.1.0¹,¹⁰.0³,⁸]tetradecan-2-yl (2E)-2-methylbut-2-enoate |
|---|
| SMILES | C\C=C(/C)C(=O)OC1C23OC2(CC2CCC(O)C(C)C12C)OC(=O)C3C |
|---|
| InChI Identifier | InChI=1S/C20H28O6/c1-6-10(2)15(22)24-17-18(5)11(3)14(21)8-7-13(18)9-19-20(17,26-19)12(4)16(23)25-19/h6,11-14,17,21H,7-9H2,1-5H3/b10-6+ |
|---|
| InChI Key | FYNKIYMILKEXOG-UXBLZVDNSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as naphthofurans. Naphthofurans are compounds containing a furan ring fused to a naphthalene moiety. Furan is a 5 membered- ring aromatic ring with four carbon and one oxygen atoms. Naphthalene is a polycyclic aromatic hydrocarbon made up of two fused benzene rings. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Naphthofurans |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Naphthofurans |
|---|
| Alternative Parents | |
|---|
| Substituents | - Naphthofuran
- Enol ester epoxide
- Fatty acid ester
- Oxepane
- Meta-dioxane
- Dicarboxylic acid or derivatives
- Gamma butyrolactone
- Fatty acyl
- Cyclic alcohol
- Alpha,beta-unsaturated carboxylic ester
- Enoate ester
- Tetrahydrofuran
- Lactone
- Carboxylic acid ester
- Secondary alcohol
- Oxacycle
- Oxirane
- Carboxylic acid derivative
- Alcohol
- Hydrocarbon derivative
- Organic oxide
- Organic oxygen compound
- Organooxygen compound
- Carbonyl group
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-000i-8797000000-eead7ceb68d171eab04d | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-05aj-9747300000-b1e5bfae56c8e6c04ad8 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014j-2039000000-2567f24b09ff33160f8e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-05o1-9133000000-23fbe9410be5cda67baa | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0ue9-9210000000-b0ae853ba0da1a9d8ef6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-2019000000-3df6e43252cd824dd343 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-01ot-6039000000-004f596d4ff74198a090 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4j-9030000000-1a0c6139c426b5248645 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014j-0009000000-aa8335a4529a260c5922 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014j-0294000000-cf1d77e675988f32957b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0aou-9325000000-40ac15a14b9b05739d0d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-2009000000-4d334d5519387a3e1a7a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03dj-5094000000-fc6d75e4546e56ea9c52 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0zfr-9050000000-d9ebcb5c5ea986c378a0 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0030899 |
|---|
| FooDB ID | FDB002863 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 35013281 |
|---|
| ChEBI ID | 175667 |
|---|
| PubChem Compound ID | 131751093 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|