| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:56:07 UTC |
|---|
| Update Date | 2016-11-09 01:17:57 UTC |
|---|
| Accession Number | CHEM024802 |
|---|
| Identification |
|---|
| Common Name | (R)-Marmin |
|---|
| Class | Small Molecule |
|---|
| Description | (R)-Marmin is found in citrus. (R)-Marmin is a constituent of grapefruit peel and bael fruit (Aegle marmelos) |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
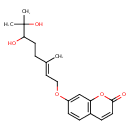
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 7-(6',7'-Dihydroxygeranyloxy)coumarin | HMDB | | Marmin | HMDB |
|
|---|
| Chemical Formula | C19H24O5 |
|---|
| Average Molecular Mass | 332.391 g/mol |
|---|
| Monoisotopic Mass | 332.162 g/mol |
|---|
| CAS Registry Number | 14957-38-1 |
|---|
| IUPAC Name | 7-{[(2E)-6,7-dihydroxy-3,7-dimethyloct-2-en-1-yl]oxy}-2H-chromen-2-one |
|---|
| Traditional Name | 7-{[(2E)-6,7-dihydroxy-3,7-dimethyloct-2-en-1-yl]oxy}chromen-2-one |
|---|
| SMILES | C\C(CCC(O)C(C)(C)O)=C/COC1=CC2=C(C=CC(=O)O2)C=C1 |
|---|
| InChI Identifier | InChI=1S/C19H24O5/c1-13(4-8-17(20)19(2,3)22)10-11-23-15-7-5-14-6-9-18(21)24-16(14)12-15/h5-7,9-10,12,17,20,22H,4,8,11H2,1-3H3/b13-10+ |
|---|
| InChI Key | QYYKWTUUCOTGNS-JLHYYAGUSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as coumarins and derivatives. These are polycyclic aromatic compounds containing a 1-benzopyran moiety with a ketone group at the C2 carbon atom (1-benzopyran-2-one). |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Coumarins and derivatives |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Coumarins and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Coumarin
- Benzopyran
- 1-benzopyran
- Fatty alcohol
- Alkyl aryl ether
- Pyranone
- Pyran
- Fatty acyl
- Benzenoid
- Tertiary alcohol
- Heteroaromatic compound
- Secondary alcohol
- 1,2-diol
- Lactone
- Oxacycle
- Ether
- Organoheterocyclic compound
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxide
- Alcohol
- Organic oxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0a4i-9143000000-b5ff0db4041e7dc062eb | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-01q9-7945800000-8c9e650383310252c022 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00lr-0449000000-8f29fd82e4fbc34a65b0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0ho4-4953000000-cbdfa7bea484157cac74 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-029i-9510000000-10ae74648a914da7ce39 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-01q9-0339000000-749a91054b580eab2a7f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-0922000000-2f3a91d3a9f1feaafea2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-014i-2900000000-f84d9eca03ab7d47cc7b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00kb-0169000000-be0c48ffd91e02a405b3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01ot-2791000000-6baf219959ed30ec432c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-03yi-9720000000-66bddcd59fad62417f43 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03e9-0908000000-8caa6681bef0b936a33b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-2910000000-0423cce82b9ab500649f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0159-0900000000-33d87450be29b1aedac5 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0030787 |
|---|
| FooDB ID | FDB002724 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00037483 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 4764935 |
|---|
| ChEBI ID | 172558 |
|---|
| PubChem Compound ID | 5964600 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|