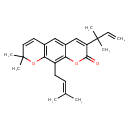| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:53:48 UTC |
|---|
| Update Date | 2016-11-09 01:17:57 UTC |
|---|
| Accession Number | CHEM024742 |
|---|
| Identification |
|---|
| Common Name | 3-(1,1-Dimethyl-2-propenyl)-8-(3-methyl-2-butenyl)xanthyletin |
|---|
| Class | Small Molecule |
|---|
| Description | 3-(1,1-Dimethyl-2-propenyl)-8-(3-methyl-2-butenyl)xanthyletin is found in herbs and spices. 3-(1,1-Dimethyl-2-propenyl)-8-(3-methyl-2-butenyl)xanthyletin is isolated from roots of Ruta graveolens (rue |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3-(1,1-Dimethylallyl)-8-(3,3-dimethylallyl)xanthyletin | HMDB |
|
|---|
| Chemical Formula | C24H28O3 |
|---|
| Average Molecular Mass | 364.477 g/mol |
|---|
| Monoisotopic Mass | 364.204 g/mol |
|---|
| CAS Registry Number | 30310-55-5 |
|---|
| IUPAC Name | 8,8-dimethyl-10-(3-methylbut-2-en-1-yl)-3-(2-methylbut-3-en-2-yl)-2H,8H-pyrano[3,2-g]chromen-2-one |
|---|
| Traditional Name | 8,8-dimethyl-10-(3-methylbut-2-en-1-yl)-3-(2-methylbut-3-en-2-yl)pyrano[3,2-g]chromen-2-one |
|---|
| SMILES | CC(C)=CCC1=C2OC(=O)C(=CC2=CC2=C1OC(C)(C)C=C2)C(C)(C)C=C |
|---|
| InChI Identifier | InChI=1S/C24H28O3/c1-8-23(4,5)19-14-17-13-16-11-12-24(6,7)27-21(16)18(10-9-15(2)3)20(17)26-22(19)25/h8-9,11-14H,1,10H2,2-7H3 |
|---|
| InChI Key | PIOKBHKPGZHPHS-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as linear pyranocoumarins. These are organic compounds containing a pyran (or a hydrogenated derivative) linearly fused to a coumarin moiety. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Coumarins and derivatives |
|---|
| Sub Class | Pyranocoumarins |
|---|
| Direct Parent | Linear pyranocoumarins |
|---|
| Alternative Parents | |
|---|
| Substituents | - Linear pyranocoumarin
- Pyranochromene
- 2,2-dimethyl-1-benzopyran
- Benzopyran
- 1-benzopyran
- Alkyl aryl ether
- Pyranone
- Pyran
- Benzenoid
- Heteroaromatic compound
- Lactone
- Oxacycle
- Ether
- Organoheterocyclic compound
- Hydrocarbon derivative
- Organooxygen compound
- Organic oxygen compound
- Organic oxide
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-052b-2029000000-11e448126c2baf34ab60 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-1019000000-a5325a38f897a5ea3022 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-066r-3029000000-77e4055c7a0c201d549e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-014i-9084000000-a311ba203e727f66c071 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0019000000-c6dadfe61d40c2ff81d6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-0049000000-e591a6c2e9c4f7384225 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-014j-4093000000-856a3d4eacb4e6c91f26 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-066r-0029000000-1dd0dc5913eddf8d7e0c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00lr-3069000000-e6a27f7a34ae07d5d5c7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0694-3094000000-aaa95ed4437973325630 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0009000000-dc6ab8188c3cddd5242c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-0019000000-550ce1f7630612b7e683 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0fc0-0092000000-1a6a0e8197e2857c5c7a | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0030730 |
|---|
| FooDB ID | FDB002656 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 10297786 |
|---|
| ChEBI ID | 174872 |
|---|
| PubChem Compound ID | 14604074 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|