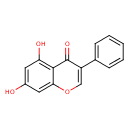
| Description | 5,7-Dihydroxyisoflavone is found in nuts. 5,7-Dihydroxyisoflavone is isolated from hydrolysed flour of Arachis hypogaea (peanut) Mefenamic acid, an anthranilic acid derivative, is a member of the fenamate group of nonsteroidal anti-inflammatory drugs (NSAIDs). It exhibits anti-inflammatory, analgesic, and antipyretic activities. Similar to other NSAIDs, mefenamic acid inhibits prostaglandin synthetase |
|---|
| Synonyms | | Value | Source |
|---|
| 2',3'-Dimethyl-2-diphenylaminecarboxylic acid | HMDB | | 2-((2,3-Dimethylphenyl)amino)-benzoic acid | HMDB | | 2-((2,3-Dimethylphenyl)amino)benzoic acid | HMDB | | 2-(2,3-dimethylanilino)Benzoic acid | HMDB | | 2-(2,3-Dimethylphenyl)amino-benzoic acid | HMDB | | 2-[(2,3-Dimethylphenyl)amino]-benzoic acid | HMDB | | 2-[(2,3-Dimethylphenyl)amino]benzoic acid | HMDB | | 5,7-Dihydroxy-3-phenyl-4H-1-benzopyran-4-one, 9ci | HMDB | | Ac. mefenamico | HMDB | | Acid, mefenamic | HMDB, MeSH | | Acid, mefenaminic | HMDB | | acido Mefenamico | HMDB | | Acidum mefenamicum | HMDB | | Antigen brand OF mefenamic acid | HMDB | | apo Mefenamic | HMDB, MeSH | | apo-Mefenamic | HMDB, MeSH | | Apomefenamic | HMDB | | Apotex brand OF mefenamic acid | HMDB | | Aps brand OF mefenamic acid | HMDB | | Ashbourne brand OF mefenamic acid | HMDB | | Bafameritin-m | HMDB | | Bafhameritin-m | HMDB | | Bonabol | HMDB | | Chemidex brand OF mefenamic acid | HMDB | | Clonmel brand OF mefenamic acid | HMDB | | Contraflam | HMDB, MeSH | | Coslan | HMDB, MeSH | | Dysman | HMDB, MeSH | | Elan brand OF mefenamic acid | HMDB | | Farmasierra brand OF mefenamic acid | HMDB | | Fenamin | HMDB | | First horizon brand OF mefenamic acid | HMDB | | Forte, ponstan | HMDB | | Lysalgo | HMDB | | Mefac | HMDB, MeSH | | Mefacit | HMDB, MeSH | | Mefedolo | HMDB | | Mefenamate | HMDB | | Mefenamic acid | HMDB, MeSH | | Mefenamic acid (JP15/usp/inn) | HMDB | | Mefenaminic acid | HMDB, MeSH | | Mefic | HMDB, MeSH | | Mephenamic acid | HMDB | | Mephenaminic acid | HMDB | | Methenamic acid | HMDB | | Mycasaal | HMDB | | N-(2, 3-Dimethylphenyl)anthranilic acid | HMDB | | N-(2,3-Dimethylphenyl)anthranilic acid | HMDB | | N-(2,3-Xylyl)-2-aminobenzoic acid | HMDB | | N-(2,3-Xylyl)-anthranilic acid | HMDB | | N-(2,3-Xylyl)anthranilic acid | HMDB | | N-2,3-Xylyl-anthranilic acid | HMDB | | N-2,3-Xylylanthranilic acid | HMDB | | Namphen | HMDB | | Nu mefenamic | HMDB, MeSH | | Nu pharm brand OF mefenamic acid | HMDB | | Nu-mefenamic | HMDB, MeSH | | Nu-pharm brand OF mefenamic acid | HMDB | | Numefenamic | HMDB | | Parke davis brand OF mefenamic acid | HMDB | | Parkemed | HMDB, MeSH | | Pfizer brand OF mefenamic acid | HMDB | | Pharmascience brand OF mefenamic acid | HMDB | | Pinalgesic | HMDB, MeSH | | Pinewood brand OF mefenamic acid | HMDB | | PMS Mefenamic acid | HMDB, MeSH | | PMS-Mefenamic acid | HMDB, MeSH | | Ponalar | HMDB, MeSH | | Ponalgic | HMDB, MeSH | | Ponmel | HMDB, MeSH | | Ponstan | HMDB, MeSH | | Ponstan forte | HMDB, MeSH | | Ponstel | HMDB, MeSH | | Ponstil | HMDB | | Ponstyl | HMDB | | Ponsyl | HMDB, MeSH | | Pontal | HMDB, MeSH | | Rolan | HMDB | | Rowa brand OF mefenamic acid | HMDB | | Tamany bonsan | HMDB | | Tanston | HMDB | | Vialidon | HMDB | | Warner lambert brand OF mefenamic acid | HMDB | | Warner-lambert brand OF mefenamic acid | HMDB |
|
|---|