| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:51:59 UTC |
|---|
| Update Date | 2016-11-09 01:17:56 UTC |
|---|
| Accession Number | CHEM024696 |
|---|
| Identification |
|---|
| Common Name | Cycloheterophyllin |
|---|
| Class | Small Molecule |
|---|
| Description | Cycloheterophyllin is found in fruits. Cycloheterophyllin is isolated from bark of Artocarpus heterophyllus (jackfruit |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
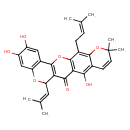
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 2,3,8-Trihydroxy-11,11-dimethyl-13-(3-methyl-2-butenyl)-6-(2-methyl-1-propenyl)-6H,7H,11H-bis[1]benzopyrano[4,3-b:6',7'-e]pyran-7-one, 9ci | HMDB |
|
|---|
| Chemical Formula | C30H30O7 |
|---|
| Average Molecular Mass | 502.555 g/mol |
|---|
| Monoisotopic Mass | 502.199 g/mol |
|---|
| CAS Registry Number | 36545-53-6 |
|---|
| IUPAC Name | 6,7,15-trihydroxy-19,19-dimethyl-22-(3-methylbut-2-en-1-yl)-11-(2-methylprop-1-en-1-yl)-2,10,20-trioxapentacyclo[12.8.0.0³,¹².0⁴,⁹.0¹⁶,²¹]docosa-1(14),3(12),4(9),5,7,15,17,21-octaen-13-one |
|---|
| Traditional Name | cycloheterophyllin |
|---|
| SMILES | CC(C)=CCC1=C2OC(C)(C)C=CC2=C(O)C2=C1OC1=C(C(OC3=C1C=C(O)C(O)=C3)C=C(C)C)C2=O |
|---|
| InChI Identifier | InChI=1S/C30H30O7/c1-14(2)7-8-17-27-16(9-10-30(5,6)37-27)25(33)24-26(34)23-22(11-15(3)4)35-21-13-20(32)19(31)12-18(21)29(23)36-28(17)24/h7,9-13,22,31-33H,8H2,1-6H3 |
|---|
| InChI Key | ZZPIXEJZTXAVCX-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as pyranoflavonoids. Pyranoflavonoids are compounds containing a pyran ring fused to a 2-phenyl-1,4-benzopyran skeleton. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Flavonoids |
|---|
| Sub Class | Pyranoflavonoids |
|---|
| Direct Parent | Pyranoflavonoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Pyranoflavonoid
- Pyranochromene
- 2,2-dimethyl-1-benzopyran
- Chromone
- Benzopyran
- 1-benzopyran
- 1-hydroxy-2-unsubstituted benzenoid
- Alkyl aryl ether
- Pyranone
- Pyran
- Benzenoid
- Heteroaromatic compound
- Vinylogous acid
- Ether
- Polyol
- Oxacycle
- Organoheterocyclic compound
- Organic oxygen compound
- Organooxygen compound
- Organic oxide
- Hydrocarbon derivative
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-000i-1210900000-2b9348571f9c2ede2c21 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-001i-2100029000-1a10b698dc6f89f727b5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-1301890000-3a9668040559528c86dc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-07bb-3103910000-3e34e7b9d27e80266f15 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0pxr-9501100000-7f051663aa9661ab0611 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0000190000-684986bbcc1af6b7bb2b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-1002960000-ab6c73fca49f7aae21e8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0apj-1445900000-e6a2476407cfa534cc22 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0000090000-27a32bd6c97a965edc44 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-0000090000-27a32bd6c97a965edc44 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4r-0090010000-7e9d5fac0bdd16aa0613 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0000090000-db49500cd3a1fa636786 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udi-0000090000-db49500cd3a1fa636786 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0f79-0090250000-7dd066fbdb79c38212a8 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0030685 |
|---|
| FooDB ID | FDB002604 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00004064 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 4475345 |
|---|
| ChEBI ID | 563917 |
|---|
| PubChem Compound ID | 5316250 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | | 1. Lin CN, Lu CM, Lin HC, Fang SC, Shieh BJ, Hsu MF, Wang JP, Ko FN, Teng CM: Novel antiplatelet constituents from formosan moraceous plants. J Nat Prod. 1996 Sep;59(9):834-8. | | 2. Yannai, Shmuel. (2004) Dictionary of food compounds with CD-ROM: Additives, flavors, and ingredients. Boca Raton: Chapman & Hall/CRC. |
|
|---|