| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:48:21 UTC |
|---|
| Update Date | 2016-11-09 01:17:55 UTC |
|---|
| Accession Number | CHEM024597 |
|---|
| Identification |
|---|
| Common Name | Dihydro-O-methylsterigmatocystin |
|---|
| Class | Small Molecule |
|---|
| Description | Dihydro-O-methylsterigmatocystin is a mycotoxin from Aspergillus flavu |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
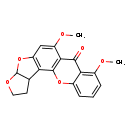
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 1,3,5,6-Tetrahydroxy-4-(3-methyl-2-butenyl)-9H-xanthen-9-one, 9ci | HMDB | | 1,2-Dihydro-6-methoxy-7-hydroxydifuroxanthone | HMDB | | 1,2-Dihydro-O-methylsterigmatocystin | HMDB | | Dhomst | HMDB |
|
|---|
| Chemical Formula | C19H16O6 |
|---|
| Average Molecular Mass | 340.327 g/mol |
|---|
| Monoisotopic Mass | 340.095 g/mol |
|---|
| CAS Registry Number | 21793-91-9 |
|---|
| IUPAC Name | 11,15-dimethoxy-6,8,20-trioxapentacyclo[10.8.0.0²,⁹.0³,⁷.0¹⁴,¹⁹]icosa-1(12),2(9),10,14,16,18-hexaen-13-one |
|---|
| Traditional Name | 11,15-dimethoxy-6,8,20-trioxapentacyclo[10.8.0.0²,⁹.0³,⁷.0¹⁴,¹⁹]icosa-1(12),2(9),10,14,16,18-hexaen-13-one |
|---|
| SMILES | COC1=CC2=C(C3CCOC3O2)C2=C1C(=O)C1=C(OC)C=CC=C1O2 |
|---|
| InChI Identifier | InChI=1S/C19H16O6/c1-21-10-4-3-5-11-15(10)17(20)16-12(22-2)8-13-14(18(16)24-11)9-6-7-23-19(9)25-13/h3-5,8-9,19H,6-7H2,1-2H3 |
|---|
| InChI Key | WSBZDTWHZJNGKQ-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as sterigmatocystins. These are a group of closely related fungal metabolites chemically characterized by a xanthone moiety fused to a dihydrodifurano or a tetrahydrodifurano moiety. The chemical difference among the various sterigmagocystins are the presence or absence of unsaturation at positions 2 and 3 of the difurano ring system, the substitution pattern on positions 6, 7, and 10 of the xanthone system and/or the substituent on position 3 of the difurano system. They are produced by Aspergilus spp. and Bipolaris spp. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Sterigmatocystins |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Sterigmatocystins |
|---|
| Alternative Parents | |
|---|
| Substituents | - Sterigmatocystin backbone
- Xanthone
- Dibenzopyran
- Xanthene
- Chromone
- Benzopyran
- 1-benzopyran
- Coumaran
- Anisole
- Alkyl aryl ether
- Pyranone
- Pyran
- Benzenoid
- Heteroaromatic compound
- Vinylogous ester
- Tetrahydrofuran
- Ether
- Oxacycle
- Acetal
- Organoheterocyclic compound
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxygen compound
- Organic oxide
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-01ot-1089000000-dbbe1612a4d70b6c8043 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0009000000-f7c69bba6f3116d91b4d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0006-0019000000-35fc0749cf7c76fbc7ed | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-1592000000-f08c2eeb7fb57efe4e73 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0009000000-dc279eb8d603e99ce625 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-000i-0019000000-716e9cd7906759337cae | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0as4-1292000000-8c9f39baa0e7fa1ecaba | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0009000000-25a309d116f84167676b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0006-0009000000-537b703ab31b5490ead8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-054k-0379000000-c73d51d05d0afe21f950 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0009000000-01d0ed7d465a91bcc831 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-052r-0019000000-cdc8266f01f4f9083606 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-056r-1796000000-20fb5aa363d89a6bf3f4 | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0030591 |
|---|
| FooDB ID | FDB002484 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00037969 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 4479504 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 119251 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|