| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:47:47 UTC |
|---|
| Update Date | 2016-11-09 01:17:55 UTC |
|---|
| Accession Number | CHEM024580 |
|---|
| Identification |
|---|
| Common Name | Tetramethylscutellarein |
|---|
| Class | Small Molecule |
|---|
| Description | A tetramethoxyflavone that is the tetra-O-methyl derivative of scutellarein. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
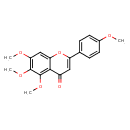
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Tetra-O-methylscutellarein | ChEBI | | 4', 5,6,7-Tetramethoxyflavone | HMDB | | 4',5,6,7-Tetramethoxy-flavone | HMDB | | 5,6,7,4'-Tetramethoxyflavone | HMDB | | 5,6,7-Trimethoxy-2-(4-methoxyphenyl)-4H-1-benzopyran-4-one | HMDB | | 5,6,7-Trimethoxy-2-(4-methoxyphenyl)-4H-chromen-4-one | HMDB | | Flavone, 4',5,6,7-tetramethoxy- (8ci) | HMDB | | Flavone, 5,6,7,4'-tetramethoxy | HMDB | | Scutellarein 5,6,7,4'-tetramethyl ether | HMDB | | Scutellarein tetramethyl ether | HMDB | | Scutellarein tetramethylether | HMDB | | Tetramethyl-O-scutellarin | HMDB | | Tetramethylscutellarein | HMDB | | 4',5,6,7-Tetramethoxyflavone | MeSH | | 4’,5,6,7-Tetramethoxyflavone | HMDB | | 5,6,7,4’-Tetramethoxyflavone | HMDB | | 5-Methoxysalvigenin | HMDB | | Pectolinarigenin dimethyl ether | HMDB |
|
|---|
| Chemical Formula | C19H18O6 |
|---|
| Average Molecular Mass | 342.343 g/mol |
|---|
| Monoisotopic Mass | 342.110 g/mol |
|---|
| CAS Registry Number | 1168-42-9 |
|---|
| IUPAC Name | 5,6,7-trimethoxy-2-(4-methoxyphenyl)-4H-chromen-4-one |
|---|
| Traditional Name | 4',5,6,7-tetramethoxyflavone |
|---|
| SMILES | COC1=CC=C(C=C1)C1=CC(=O)C2=C(OC)C(OC)=C(OC)C=C2O1 |
|---|
| InChI Identifier | InChI=1S/C19H18O6/c1-21-12-7-5-11(6-8-12)14-9-13(20)17-15(25-14)10-16(22-2)18(23-3)19(17)24-4/h5-10H,1-4H3 |
|---|
| InChI Key | URSUMOWUGDXZHU-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as 7-o-methylated flavonoids. These are flavonoids with methoxy groups attached to the C7 atom of the flavonoid backbone. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Flavonoids |
|---|
| Sub Class | O-methylated flavonoids |
|---|
| Direct Parent | 7-O-methylated flavonoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - 4p-methoxyflavonoid-skeleton
- 5-methoxyflavonoid-skeleton
- 6-methoxyflavonoid-skeleton
- 7-methoxyflavonoid-skeleton
- Flavone
- Chromone
- Benzopyran
- 1-benzopyran
- Phenoxy compound
- Methoxybenzene
- Anisole
- Phenol ether
- Pyranone
- Alkyl aryl ether
- Monocyclic benzene moiety
- Pyran
- Benzenoid
- Heteroaromatic compound
- Vinylogous ester
- Oxacycle
- Organoheterocyclic compound
- Ether
- Organic oxide
- Organic oxygen compound
- Organooxygen compound
- Hydrocarbon derivative
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-03di-0549000000-36230e769480442bc7e0 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-qTof , Positive | splash10-00di-0901000000-92a507468e91bbb0f99f | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - , positive | splash10-03ea-0696000000-2a2036cf1aa52e145023 | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - , positive | splash10-01ox-0369000000-38efb629b1fb86a82bb8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0009000000-378d17496375ca581aa0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0006-0009000000-1c1bec467a4824109868 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-03e9-1494000000-282dead7c6482eb9cf13 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0009000000-ccd544873ea637418bc5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-0009000000-498bca5d53bf1812d46a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-057i-1391000000-d2ee0102f8e8757b6f0a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0009000000-c092851bf08ad86bb4d5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-002g-0049000000-72cca8335478dc91539f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0009000000-64ce196904b844e80563 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0006-0009000000-b218ce495b244b76557b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0ufs-0369000000-3a05341a85bcb9bd8891 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0030575 |
|---|
| FooDB ID | FDB002465 |
|---|
| Phenol Explorer ID | 244 |
|---|
| KNApSAcK ID | C00003841 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 86762 |
|---|
| ChEBI ID | 34357 |
|---|
| PubChem Compound ID | 96118 |
|---|
| Kegg Compound ID | C14472 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|