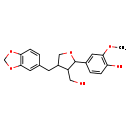| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:47:39 UTC |
|---|
| Update Date | 2016-11-09 01:17:55 UTC |
|---|
| Accession Number | CHEM024576 |
|---|
| Identification |
|---|
| Common Name | Sanshodiol |
|---|
| Class | Small Molecule |
|---|
| Description | Sanshodiol is found in herbs and spices. Sanshodiol is isolated from bark of Zanthoxylum piperitum (Japanese pepper tree |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (5-Chloro-2-hydroxyphenyl)acetic acid | HMDB | | 4,9-Dihydroxy-3-methoxy-3',4'-methylenedioxy-7,9'-epoxylignan | HMDB |
|
|---|
| Chemical Formula | C20H22O6 |
|---|
| Average Molecular Mass | 358.385 g/mol |
|---|
| Monoisotopic Mass | 358.142 g/mol |
|---|
| CAS Registry Number | 54854-91-0 |
|---|
| IUPAC Name | 4-[4-(2H-1,3-benzodioxol-5-ylmethyl)-3-(hydroxymethyl)oxolan-2-yl]-2-methoxyphenol |
|---|
| Traditional Name | 4-[4-(2H-1,3-benzodioxol-5-ylmethyl)-3-(hydroxymethyl)oxolan-2-yl]-2-methoxyphenol |
|---|
| SMILES | COC1=C(O)C=CC(=C1)C1OCC(CC2=CC3=C(OCO3)C=C2)C1CO |
|---|
| InChI Identifier | InChI=1S/C20H22O6/c1-23-18-8-13(3-4-16(18)22)20-15(9-21)14(10-24-20)6-12-2-5-17-19(7-12)26-11-25-17/h2-5,7-8,14-15,20-22H,6,9-11H2,1H3 |
|---|
| InChI Key | GRYMYKQGSSTJBA-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as 7,9'-epoxylignans. These are lignans that contain the 7,9'-epoxylignan skeleton, which consists of a tetrahydrofuran that carries a phenyl group, a methyl group, and a benzyl group at the 2-, 3-, 4-position, respectively. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lignans, neolignans and related compounds |
|---|
| Class | Furanoid lignans |
|---|
| Sub Class | Tetrahydrofuran lignans |
|---|
| Direct Parent | 7,9'-epoxylignans |
|---|
| Alternative Parents | |
|---|
| Substituents | - 7,9p-epoxylignan
- Methoxyphenol
- Benzodioxole
- Anisole
- Phenoxy compound
- Phenol ether
- Methoxybenzene
- 1-hydroxy-2-unsubstituted benzenoid
- Phenol
- Alkyl aryl ether
- Benzenoid
- Monocyclic benzene moiety
- Tetrahydrofuran
- Oxacycle
- Acetal
- Organoheterocyclic compound
- Dialkyl ether
- Ether
- Organic oxygen compound
- Alcohol
- Hydrocarbon derivative
- Primary alcohol
- Organooxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-002r-0915000000-c65b502778405c67b0b8 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-0079-3614900000-decff1b683248fcdb020 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4l-0119000000-0973db979ab10f8cd4b4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-052f-0479000000-5a297bf64bddd8311db0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-01r6-1900000000-d697d07bd006fa5fb3a5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0009000000-1bbfab0937ac8c3b7c4d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a70-0019000000-2c0e7d81516c2f2271c8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0h2u-2922000000-bd059dcc8fe5d185716c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0109000000-8fd4cfea26a15698db1f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-052r-0849000000-3231d0db4af7fd99ac1c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-000i-0922000000-9bed0b5b21b563e0a95d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a6r-0009000000-3a9c7b03e710a983f95a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a6r-0129000000-f9b40bb6aea1ce08318d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-2439000000-6d6c0b73c8d7b52f8706 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0030573 |
|---|
| FooDB ID | FDB002461 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00057434 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 35013228 |
|---|
| ChEBI ID | 175604 |
|---|
| PubChem Compound ID | 14237706 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|