| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:40:26 UTC |
|---|
| Update Date | 2016-11-09 01:17:53 UTC |
|---|
| Accession Number | CHEM024396 |
|---|
| Identification |
|---|
| Common Name | Maltoxazine |
|---|
| Class | Small Molecule |
|---|
| Description | Aroma substance isolated from malt. Alkaloid from fruit of Chinese jujube Zizyphus jujuba variety inermis (Rhamnaceae). Maltoxazine is found in cereals and cereal products and fruits. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
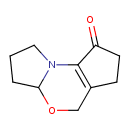
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 1,2,3,3a,6,7-Hexahydrocyclopenta[D]pyrrolo[2,1-b][1,3]oxazin-8(5H)-one, 9ci | HMDB | | 8-oxo-1,2,3,3a,5,6,7,8-Octahydrocyclopenta[D]pyrrolo[2,1-b][1,3]oxazine | HMDB | | Daechualkaloid a | HMDB |
|
|---|
| Chemical Formula | C10H13NO2 |
|---|
| Average Molecular Mass | 179.216 g/mol |
|---|
| Monoisotopic Mass | 179.095 g/mol |
|---|
| CAS Registry Number | 80933-73-9 |
|---|
| IUPAC Name | 8-oxa-1-azatricyclo[7.3.0.0²,⁶]dodec-2(6)-en-3-one |
|---|
| Traditional Name | 8-oxa-1-azatricyclo[7.3.0.0²,⁶]dodec-2(6)-en-3-one |
|---|
| SMILES | O=C1CCC2=C1N1CCCC1OC2 |
|---|
| InChI Identifier | InChI=1S/C10H13NO2/c12-8-4-3-7-6-13-9-2-1-5-11(9)10(7)8/h9H,1-6H2 |
|---|
| InChI Key | MTHASAHNRVFFOM-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as pyrrolidines. Pyrrolidines are compounds containing a pyrrolidine ring, which is a five-membered saturated aliphatic heterocycle with one nitrogen atom and four carbon atoms. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Pyrrolidines |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Pyrrolidines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Pyrrolidine
- Ketone
- Oxacycle
- Azacycle
- Enamine
- Organic nitrogen compound
- Organic oxygen compound
- Organopnictogen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Organonitrogen compound
- Carbonyl group
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0fr6-9800000000-5154de51f34bf2f4b506 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-0900000000-2c42dff9c089d137c0eb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-0900000000-71c47d26144cb854a3cc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00r6-9100000000-f90c99a9768c4c822019 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0900000000-81ae423111b56f7ec480 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-001i-9400000000-794a2b395bde851ba7ef | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0aor-9200000000-e436f2d6f6d2c99d3002 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0900000000-e9e4d0ba83478e578c42 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-004i-0900000000-8df8e7bfdac219838352 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0gi1-3900000000-d9bdd9a66e241a18d7b1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-0900000000-6326446584297a254a6a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-0900000000-7a75081e4798c7ab96f7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0fk9-5900000000-098b7798877b6ffaa062 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0030372 |
|---|
| FooDB ID | FDB002221 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 13875298 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|