| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:39:53 UTC |
|---|
| Update Date | 2016-11-09 01:17:53 UTC |
|---|
| Accession Number | CHEM024381 |
|---|
| Identification |
|---|
| Common Name | N-Methylaspidospermatidine |
|---|
| Class | Small Molecule |
|---|
| Description | Alkaloid in Aspidosperma quebracho-blanco (quebracho) |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
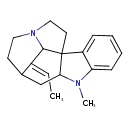
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 14,19-Didehydro-1-methyl-(14E)-condyfolan | HMDB |
|
|---|
| Chemical Formula | C19H24N2 |
|---|
| Average Molecular Mass | 280.407 g/mol |
|---|
| Monoisotopic Mass | 280.194 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | (18E)-18-ethylidene-8-methyl-8,14-diazapentacyclo[9.5.2.0¹,⁹.0²,⁷.0¹⁴,¹⁷]octadeca-2,4,6-triene |
|---|
| Traditional Name | (18E)-18-ethylidene-8-methyl-8,14-diazapentacyclo[9.5.2.0¹,⁹.0²,⁷.0¹⁴,¹⁷]octadeca-2,4,6-triene |
|---|
| SMILES | C\C=C1\C2N3CCC22C(CC1CC3)N(C)C1=CC=CC=C21 |
|---|
| InChI Identifier | InChI=1S/C19H24N2/c1-3-14-13-8-10-21-11-9-19(18(14)21)15-6-4-5-7-16(15)20(2)17(19)12-13/h3-7,13,17-18H,8-12H2,1-2H3/b14-3+ |
|---|
| InChI Key | VYHOVEWQRGHTOX-LZWSPWQCSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as strychnos alkaloids. These are alkaloids having a core structure based on the strychnan, stemmadenine (seco-curan), or the akuammicine (curan) skeleton. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Alkaloids and derivatives |
|---|
| Class | Strychnos alkaloids |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Strychnos alkaloids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Aspidosperma alkaloid
- Stemmadenine-skeleton
- Carbazole
- Indole or derivatives
- Indolizidine
- Dialkylarylamine
- Tertiary aliphatic/aromatic amine
- Aralkylamine
- N-alkylpyrrolidine
- Piperidine
- Benzenoid
- Pyrrolidine
- Tertiary aliphatic amine
- Tertiary amine
- Organoheterocyclic compound
- Azacycle
- Amine
- Organonitrogen compound
- Hydrocarbon derivative
- Organopnictogen compound
- Organic nitrogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0udi-0090000000-95f5fffde7b997c6c5ca | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-0090000000-b0434c3e4c1a7c222834 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-0090000000-137cd5e8a460927f8674 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0aor-1790000000-434a36377d9c4d8fc2cd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0090000000-b7e0722a0aee062a8fed | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-004i-0090000000-d9ce19d500bf0438ed36 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-03di-0090000000-496549949449878ae554 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0090000000-fa0545de4a4dc60fd062 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-004i-0090000000-fa0545de4a4dc60fd062 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-004i-0390000000-71f1a559c3ff9ed28cec | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-0090000000-633af5025278f639cee6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-0090000000-dd0e47ae3feb7221afc2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0059-0090000000-6a9e001e7f5f929cf4cd | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0030357 |
|---|
| FooDB ID | FDB002203 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 131751005 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|