| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:39:29 UTC |
|---|
| Update Date | 2016-11-09 01:17:53 UTC |
|---|
| Accession Number | CHEM024367 |
|---|
| Identification |
|---|
| Common Name | (R)-Pronuciferine |
|---|
| Class | Small Molecule |
|---|
| Description | (R)-Pronuciferine is found in cherimoya. (R)-Pronuciferine is an alkaloid from Nelumbo nucifera (East India lotus |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
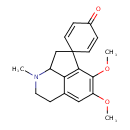
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (+)-Pronuciferine | HMDB | | Milthanthine | HMDB | | N,O-Dimethylcrotonosine | HMDB | | N-Methylstepharine | HMDB | | Pronuciferin | HMDB | | Pronuciferine | HMDB |
|
|---|
| Chemical Formula | C19H21NO3 |
|---|
| Average Molecular Mass | 311.375 g/mol |
|---|
| Monoisotopic Mass | 311.152 g/mol |
|---|
| CAS Registry Number | 2128-60-1 |
|---|
| IUPAC Name | 10',11'-dimethoxy-5'-methyl-5'-azaspiro[cyclohexane-1,2'-tricyclo[6.3.1.0⁴,¹²]dodecane]-1'(12'),2,5,8',10'-pentaen-4-one |
|---|
| Traditional Name | 10',11'-dimethoxy-5'-methyl-5'-azaspiro[cyclohexane-1,2'-tricyclo[6.3.1.0⁴,¹²]dodecane]-1'(12'),2,5,8',10'-pentaen-4-one |
|---|
| SMILES | COC1=C(OC)C2=C3C(CC22C=CC(=O)C=C2)N(C)CCC3=C1 |
|---|
| InChI Identifier | InChI=1S/C19H21NO3/c1-20-9-6-12-10-15(22-2)18(23-3)17-16(12)14(20)11-19(17)7-4-13(21)5-8-19/h4-5,7-8,10,14H,6,9,11H2,1-3H3 |
|---|
| InChI Key | WUYQEGNOQLRQAQ-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as proaporphines. These are benzylisoquinoline derivatives characterized by the presence of a spirocyclohexane ring that can occur in various oxidation levels, from cyclohexadienone to cyclohexanol. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Alkaloids and derivatives |
|---|
| Class | Proaporphines |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Proaporphines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Proaporphine
- Tetrahydroisoquinoline
- Indane
- Anisole
- Alkyl aryl ether
- Aralkylamine
- Benzenoid
- Ketone
- Tertiary amine
- Cyclic ketone
- Tertiary aliphatic amine
- Ether
- Azacycle
- Organoheterocyclic compound
- Organooxygen compound
- Organonitrogen compound
- Amine
- Hydrocarbon derivative
- Organopnictogen compound
- Carbonyl group
- Organic nitrogen compound
- Organic oxide
- Organic oxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0002-0090000000-86334b4a99135535b468 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0059000000-24ed1e33f6d78af24232 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01q9-0092000000-f4f2970614db51a81d97 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0v00-2090000000-14cc8227698e8328a57e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0009000000-35a9eef82c86df85fa6e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-0079000000-79ce4bd105b65de6f1df | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-03kc-0090000000-86e72526e0832cd9e0b8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0009000000-9f3b29c8dcdf015b99e9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03di-0039000000-5009deaf9268e5c4f8a9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0909-1090000000-41fa0a4fc39efba03adc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0009000000-9bf675b2e28db05b0d3b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-0009000000-7c0bcc885755bb30ace8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-1049000000-bd5ec9bb499964d23c7e | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0030344 |
|---|
| FooDB ID | FDB002188 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00001905 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 545754 |
|---|
| ChEBI ID | 42 |
|---|
| PubChem Compound ID | 628376 |
|---|
| Kegg Compound ID | C09611 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|