| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:38:19 UTC |
|---|
| Update Date | 2016-11-09 01:17:52 UTC |
|---|
| Accession Number | CHEM024332 |
|---|
| Identification |
|---|
| Common Name | O-Isopentenylhalfordinol |
|---|
| Class | Small Molecule |
|---|
| Description | O-Isopentenylhalfordinol is found in fruits. O-Isopentenylhalfordinol is an alkaloid from Aegle marmelos (bael fruit |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
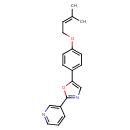
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3-[5-[4-[(3-Methyl-2-butenyl)oxy]phenyl]-2-oxazolyl]pyridine, 9ci | HMDB | | O-(3,3-Dimethylallyl)halfordinol | HMDB | | O-Prenylhalfordinol | HMDB |
|
|---|
| Chemical Formula | C19H18N2O2 |
|---|
| Average Molecular Mass | 306.358 g/mol |
|---|
| Monoisotopic Mass | 306.137 g/mol |
|---|
| CAS Registry Number | 17190-80-6 |
|---|
| IUPAC Name | 3-(5-{4-[(3-methylbut-2-en-1-yl)oxy]phenyl}-1,3-oxazol-2-yl)pyridine |
|---|
| Traditional Name | 3-(5-{4-[(3-methylbut-2-en-1-yl)oxy]phenyl}-1,3-oxazol-2-yl)pyridine |
|---|
| SMILES | CC(C)=CCOC1=CC=C(C=C1)C1=CN=C(O1)C1=CN=CC=C1 |
|---|
| InChI Identifier | InChI=1S/C19H18N2O2/c1-14(2)9-11-22-17-7-5-15(6-8-17)18-13-21-19(23-18)16-4-3-10-20-12-16/h3-10,12-13H,11H2,1-2H3 |
|---|
| InChI Key | CPMFTHYYYPZYOB-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as phenyl-1,3-oxazoles. These are aromatic heterocyclic compounds containing a 1,3-oxazole substituted at one or more positions by a phenyl group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Azoles |
|---|
| Sub Class | Oxazoles |
|---|
| Direct Parent | Phenyl-1,3-oxazoles |
|---|
| Alternative Parents | |
|---|
| Substituents | - Phenyl-1,3-oxazole
- Phenoxy compound
- Phenol ether
- 2,5-disubstituted 1,3-oxazole
- Alkyl aryl ether
- Monocyclic benzene moiety
- Pyridine
- Benzenoid
- Heteroaromatic compound
- Ether
- Oxacycle
- Azacycle
- Organooxygen compound
- Organonitrogen compound
- Organic nitrogen compound
- Organopnictogen compound
- Hydrocarbon derivative
- Organic oxygen compound
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-000f-5490000000-d02a79f4ec33bc3f8921 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-2029000000-466cb2e07c4eb4f7748a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014i-9141000000-2c41fd89da3e90d6427f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0gc0-9410000000-432c25e7ad279a637037 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0039000000-8259f7f2a86885d4765d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-000i-0091000000-d4ca34d984a68e9b7cb1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-003i-4690000000-e57eab2fa874d1b3be14 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-052r-0096000000-09195325fbcbaf3bf963 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00kr-1092000000-472034f4c28d277485ad | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-01ox-7590000000-4a841385ca477d521256 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0093000000-c0dccdd24ca7060322eb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4r-0097000000-9c04eb5dfba60ec67438 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0019-0690000000-b99d16442ac11cca36c0 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0030308 |
|---|
| FooDB ID | FDB002146 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00057139 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 536452 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 617263 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|