| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:36:58 UTC |
|---|
| Update Date | 2016-11-09 01:17:52 UTC |
|---|
| Accession Number | CHEM024297 |
|---|
| Identification |
|---|
| Common Name | Dehydrocarpaine II |
|---|
| Class | Small Molecule |
|---|
| Description | Dehydrocarpaine II is found in fruits. Dehydrocarpaine II is an alkaloid from the leaves of Carica papaya (papaya |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
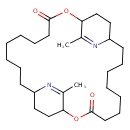
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 12,13,25,26-Tetradehydrocarpaine, 9ci | HMDB |
|
|---|
| Chemical Formula | C28H46N2O4 |
|---|
| Average Molecular Mass | 474.676 g/mol |
|---|
| Monoisotopic Mass | 474.346 g/mol |
|---|
| CAS Registry Number | 72362-03-9 |
|---|
| IUPAC Name | 13,26-dimethyl-2,15-dioxa-12,25-diazatricyclo[22.2.2.2¹¹,¹⁴]triaconta-12,25-diene-3,16-dione |
|---|
| Traditional Name | 13,26-dimethyl-2,15-dioxa-12,25-diazatricyclo[22.2.2.2¹¹,¹⁴]triaconta-12,25-diene-3,16-dione |
|---|
| SMILES | CC1=NC2CCC1OC(=O)CCCCCCCC1CCC(OC(=O)CCCCCCC2)C(C)=N1 |
|---|
| InChI Identifier | InChI=1S/C28H46N2O4/c1-21-25-19-17-23(29-21)13-9-5-3-8-12-16-28(32)34-26-20-18-24(30-22(26)2)14-10-6-4-7-11-15-27(31)33-25/h23-26H,3-20H2,1-2H3 |
|---|
| InChI Key | OEDAQCLCHGASNH-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as macrolides and analogues. These are organic compounds containing a lactone ring of at least twelve members. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Macrolides and analogues |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Macrolides and analogues |
|---|
| Alternative Parents | |
|---|
| Substituents | - Macrolide
- Tetrahydropyridine
- Dicarboxylic acid or derivatives
- Carboxylic acid ester
- Ketimine
- Lactone
- Carboxylic acid derivative
- Oxacycle
- Propargyl-type 1,3-dipolar organic compound
- Organic 1,3-dipolar compound
- Organoheterocyclic compound
- Azacycle
- Organonitrogen compound
- Hydrocarbon derivative
- Organic oxide
- Organopnictogen compound
- Organooxygen compound
- Carbonyl group
- Organic nitrogen compound
- Organic oxygen compound
- Imine
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-00di-0000900000-3accd908c9724f2c15ae | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-0000900000-8138c4a4e7d33ee30be1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-004i-0000900000-60af738bdd5e610b6cac | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a6r-0000900000-878b7ef2fec1a91f66d5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-0000900000-7b99d3ab3c913e0a1869 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00di-0000900000-0a7bfd854dd36c50f2e4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-0000900000-78cfd42aaf05a826f6b0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-0000900000-82ed194c5bbd567e1004 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-004i-0000900000-82ed194c5bbd567e1004 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a6r-0000900000-c863ab74294ba76666e5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-0000900000-980739a83b460aa7e4c7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00di-0000900000-980739a83b460aa7e4c7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-0000900000-63ab5e6de26ed3542799 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0030273 |
|---|
| FooDB ID | FDB002104 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00054513 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | MYF6 |
|---|
| Chemspider ID | 35013171 |
|---|
| ChEBI ID | 169208 |
|---|
| PubChem Compound ID | 5318023 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|