| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:36:33 UTC |
|---|
| Update Date | 2016-11-09 01:17:51 UTC |
|---|
| Accession Number | CHEM024284 |
|---|
| Identification |
|---|
| Common Name | Quinine sulfate |
|---|
| Class | Small Molecule |
|---|
| Description | Bitter flavouring
Bitter flavouring It is used in tonics and bitter drinks Quinine is a natural white crystalline alkaloid having antipyretic, antimalarial, analgesic, anti-inflammatory properties and a bitter taste. It is a stereoisomer of quinidine which, unlike quinine, is an anti-arrhythmic. Though it has been synthesized in the lab, the bark of the cinchona tree is the only known natural source of quinine. Quinine was the first effective treatment for malaria caused by Plasmodium falciparum, appearing in therapeutics in the 17th century. It remained the antimalarial drug of choice until the 1940s, when other drugs replaced it. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
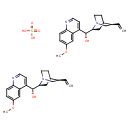
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Quinine sulfuric acid | Generator | | Quinine sulphate | Generator | | Quinine sulphuric acid | Generator |
|
|---|
| Chemical Formula | C40H50N4O8S |
|---|
| Average Molecular Mass | 746.912 g/mol |
|---|
| Monoisotopic Mass | 746.335 g/mol |
|---|
| CAS Registry Number | 804-63-7 |
|---|
| IUPAC Name | bis((R)-[(2S,5R)-5-ethenyl-1-azabicyclo[2.2.2]octan-2-yl](6-methoxyquinolin-4-yl)methanol); sulfuric acid |
|---|
| Traditional Name | bis(quinine); sulfuric acid |
|---|
| SMILES | OS(O)(=O)=O.COC1=CC=C2N=CC=C([C@@H](O)[C@@H]3CC4CCN3C[C@@H]4C=C)C2=C1.COC1=CC=C2N=CC=C([C@@H](O)[C@@H]3CC4CCN3C[C@@H]4C=C)C2=C1 |
|---|
| InChI Identifier | InChI=1S/2C20H24N2O2.H2O4S/c2*1-3-13-12-22-9-7-14(13)10-19(22)20(23)16-6-8-21-18-5-4-15(24-2)11-17(16)18;1-5(2,3)4/h2*3-6,8,11,13-14,19-20,23H,1,7,9-10,12H2,2H3;(H2,1,2,3,4)/t2*13-,14?,19-,20+;/m00./s1 |
|---|
| InChI Key | RONWGALEIBILOG-PLYVKUMQSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as cinchona alkaloids. These are alkaloids structurally characterized by the presence of the cinchonan skeleton, which consists of a quinoline linked to an azabicyclo[2.2.2]octane moiety. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Alkaloids and derivatives |
|---|
| Class | Cinchona alkaloids |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Cinchona alkaloids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Cinchonan-skeleton
- 4-quinolinemethanol
- Quinoline
- Anisole
- Quinuclidine
- Alkyl aryl ether
- Sulfuric acid
- Aralkylamine
- Benzenoid
- Pyridine
- Piperidine
- Heteroaromatic compound
- Organic sulfuric acid or derivatives
- 1,2-aminoalcohol
- Tertiary aliphatic amine
- Tertiary amine
- Secondary alcohol
- Organoheterocyclic compound
- Azacycle
- Ether
- Hydrocarbon derivative
- Organonitrogen compound
- Organooxygen compound
- Alcohol
- Organopnictogen compound
- Organic oxygen compound
- Organic nitrogen compound
- Amine
- Organic oxide
- Aromatic alcohol
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Not Available |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-0000000900-32112dd7449c53fec9c7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0002-0000000900-32112dd7449c53fec9c7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0002-0000000900-32112dd7449c53fec9c7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-0000000900-3c017f040f957dca7543 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0002-0000000900-3c017f040f957dca7543 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0002-0000000900-3c017f040f957dca7543 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0030261 |
|---|
| FooDB ID | FDB002086 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 13119 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|