| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:35:10 UTC |
|---|
| Update Date | 2016-11-09 01:17:51 UTC |
|---|
| Accession Number | CHEM024247 |
|---|
| Identification |
|---|
| Common Name | Murrayacinine |
|---|
| Class | Small Molecule |
|---|
| Description | Murrayacinine is found in herbs and spices. Murrayacinine is an alkaloid from the stem bark of Murraya koenigii (curryleaf tree |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
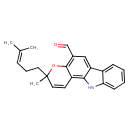
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3,11-Dihydro-3-methyl-3-(4-methyl-3-pentenyl)pyrano[3,2-a]carbazole-5-carboxaldehyde, 9ci | HMDB | | AFC | HMDB |
|
|---|
| Chemical Formula | C23H23NO2 |
|---|
| Average Molecular Mass | 345.434 g/mol |
|---|
| Monoisotopic Mass | 345.173 g/mol |
|---|
| CAS Registry Number | 53508-00-2 |
|---|
| IUPAC Name | 3-methyl-3-(4-methylpent-3-en-1-yl)-3H,11H-pyrano[3,2-a]carbazole-5-carbaldehyde |
|---|
| Traditional Name | 3-methyl-3-(4-methylpent-3-en-1-yl)-11H-pyrano[3,2-a]carbazole-5-carbaldehyde |
|---|
| SMILES | CC(C)=CCCC1(C)OC2=C(C=O)C=C3C(NC4=CC=CC=C34)=C2C=C1 |
|---|
| InChI Identifier | InChI=1S/C23H23NO2/c1-15(2)7-6-11-23(3)12-10-18-21-19(13-16(14-25)22(18)26-23)17-8-4-5-9-20(17)24-21/h4-5,7-10,12-14,24H,6,11H2,1-3H3 |
|---|
| InChI Key | VEZSSHVWEILOAM-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as carbazoles. Carbazoles are compounds containing a three ring system containing a pyrrole ring fused on either side to a benzene ring. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Indoles and derivatives |
|---|
| Sub Class | Carbazoles |
|---|
| Direct Parent | Carbazoles |
|---|
| Alternative Parents | |
|---|
| Substituents | - Carbazole
- Benzopyran
- 1-benzopyran
- Indole
- Alkyl aryl ether
- Aryl-aldehyde
- Benzenoid
- Pyrrole
- Heteroaromatic compound
- Ether
- Oxacycle
- Azacycle
- Organic nitrogen compound
- Hydrocarbon derivative
- Aldehyde
- Organooxygen compound
- Organonitrogen compound
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-00o0-9324000000-3d1d4b5c65d13299aff6 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-1029000000-582d95fd751dd6a14a15 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00di-2192000000-b0961c85f4b383710799 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0gi0-9770000000-394e509f73c27e79b07f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0019000000-5a0c8f390480980473fc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-0249000000-4d80f6b98c3ad50b7a0e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-0921000000-6e34103d66c32e196194 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0009000000-942f4b9007ac8b410396 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-0019000000-0643e84e36a237eb82ff | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-001i-1092000000-a0d022a467b005673416 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-0039000000-56af1429c08711b6b510 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-004j-0096000000-f63e9fcdb69a85aa5be4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-02ti-4090000000-f5ecace320e4deabe94b | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0030224 |
|---|
| FooDB ID | FDB002043 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00054821 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 35013163 |
|---|
| ChEBI ID | 168925 |
|---|
| PubChem Compound ID | 102219164 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|