| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:34:50 UTC |
|---|
| Update Date | 2016-11-09 01:17:51 UTC |
|---|
| Accession Number | CHEM024236 |
|---|
| Identification |
|---|
| Common Name | Mukonicine |
|---|
| Class | Small Molecule |
|---|
| Description | Mukonicine is found in herbs and spices. Mukonicine is an alkaloid from the leaves of Murraya koenigii (curry leaf tree |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
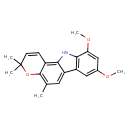
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3,11-Dihydro-8,10-dimethoxy-3,3,5-trimethylpyrano[3,2-a]carbazole, 9ci | HMDB |
|
|---|
| Chemical Formula | C20H21NO3 |
|---|
| Average Molecular Mass | 323.386 g/mol |
|---|
| Monoisotopic Mass | 323.152 g/mol |
|---|
| CAS Registry Number | 88607-43-6 |
|---|
| IUPAC Name | 13,15-dimethoxy-5,5,8-trimethyl-6-oxa-17-azatetracyclo[8.7.0.0²,⁷.0¹¹,¹⁶]heptadeca-1,3,7,9,11(16),12,14-heptaene |
|---|
| Traditional Name | 13,15-dimethoxy-5,5,8-trimethyl-6-oxa-17-azatetracyclo[8.7.0.0²,⁷.0¹¹,¹⁶]heptadeca-1,3,7,9,11(16),12,14-heptaene |
|---|
| SMILES | COC1=CC2=C(NC3=C4C=CC(C)(C)OC4=C(C)C=C23)C(OC)=C1 |
|---|
| InChI Identifier | InChI=1S/C20H21NO3/c1-11-8-14-15-9-12(22-4)10-16(23-5)18(15)21-17(14)13-6-7-20(2,3)24-19(11)13/h6-10,21H,1-5H3 |
|---|
| InChI Key | APZRRMFSHXMDTQ-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as carbazoles. Carbazoles are compounds containing a three ring system containing a pyrrole ring fused on either side to a benzene ring. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Indoles and derivatives |
|---|
| Sub Class | Carbazoles |
|---|
| Direct Parent | Carbazoles |
|---|
| Alternative Parents | |
|---|
| Substituents | - Carbazole
- 2,2-dimethyl-1-benzopyran
- Benzopyran
- 1-benzopyran
- Indole
- Anisole
- Alkyl aryl ether
- Benzenoid
- Heteroaromatic compound
- Pyrrole
- Ether
- Oxacycle
- Azacycle
- Organic oxygen compound
- Organooxygen compound
- Organonitrogen compound
- Organic nitrogen compound
- Hydrocarbon derivative
- Organopnictogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0a4i-0195000000-9b75b81b5b887d58bbc9 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00di-0029000000-faa0e8b156be4bed3136 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00dl-0094000000-108076fccbe922aee48c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-02ti-1090000000-5298a73c514a8baa764e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-0019000000-79eeb72624074c61f300 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00di-0069000000-cf51bd943f39bc12ab2f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0ka9-0090000000-5ecdc6574c8f9965c7af | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-0009000000-b8817382b3fde66270d6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00di-0009000000-131076375e7afba8b6f7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-004i-0090000000-ca9c359741560905f30f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00di-0009000000-7684723ce4870da7ab11 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00di-0039000000-0037c5d1d594a7363929 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-014i-1091000000-cc3acc7caaa6f321b9fc | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0030212 |
|---|
| FooDB ID | FDB002031 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00024719 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 30776819 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 86242003 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|