| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:33:45 UTC |
|---|
| Update Date | 2016-11-09 01:17:51 UTC |
|---|
| Accession Number | CHEM024208 |
|---|
| Identification |
|---|
| Common Name | Eupolauridine |
|---|
| Class | Small Molecule |
|---|
| Description | Eupolauridine is an alkaloid from the bark of Cananga odorata (ylang ylang |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
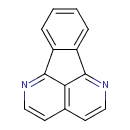
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 5-Methoxy-2-pyrrolidinone | HMDB | | 5-Methoxypyrrolidin-2-one | HMDB | | 1,6-Diazafluoranthene | HMDB | | Canangine | HMDB | | EL base 1 | HMDB | | Indeno(1,2,3-ij)(2,7)naphthyridine | HMDB | | Eupolauridine | MeSH |
|
|---|
| Chemical Formula | C14H8N2 |
|---|
| Average Molecular Mass | 204.227 g/mol |
|---|
| Monoisotopic Mass | 204.069 g/mol |
|---|
| CAS Registry Number | 58786-39-3 |
|---|
| IUPAC Name | 2,8-diazatetracyclo[7.6.1.0⁵,¹⁶.0¹⁰,¹⁵]hexadeca-1,3,5(16),6,8,10,12,14-octaene |
|---|
| Traditional Name | 2,8-diazatetracyclo[7.6.1.0⁵,¹⁶.0¹⁰,¹⁵]hexadeca-1,3,5(16),6,8,10,12,14-octaene |
|---|
| SMILES | C1=CC=C2C(=C1)C1=NC=CC3=C1C2=NC=C3 |
|---|
| InChI Identifier | InChI=1S/C14H8N2/c1-2-4-11-10(3-1)13-12-9(5-7-15-13)6-8-16-14(11)12/h1-8H |
|---|
| InChI Key | KIVUUVOREYMMFE-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as naphthyridines. Naphthyridines are compounds containing a naphthyridine moiety, a naphthalene in which a carbon atom has been replaced by a nitrogen in each of the two rings. The naphthyridine skeleton can also be described as an assembly two fused pyridine rings, which do not share their nitrogen atom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Diazanaphthalenes |
|---|
| Sub Class | Naphthyridines |
|---|
| Direct Parent | Naphthyridines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Naphthyridine
- Benzenoid
- Pyridine
- Heteroaromatic compound
- Azacycle
- Organic nitrogen compound
- Organopnictogen compound
- Hydrocarbon derivative
- Organonitrogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0udi-0590000000-fff666abdc21567cc8b4 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0090000000-339f07497a9b5b6e4a5d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-0090000000-6957b7b654f953dabb8f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4i-1390000000-7c6bb2cfd572a1b65e56 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0090000000-a069f2057d0e6cb6835f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-0090000000-a069f2057d0e6cb6835f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0udi-0290000000-a26c8ae1a054fea18e65 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0090000000-293c12776392e6a1fa6f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-0090000000-293c12776392e6a1fa6f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a6r-0690000000-aae729eec62f037510c4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0090000000-ce5af2342e5716a3bc9e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-0090000000-ce5af2342e5716a3bc9e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0udi-0090000000-ce5af2342e5716a3bc9e | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0030182 |
|---|
| FooDB ID | FDB001998 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00054236 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 65398 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 181561 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|