| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:32:52 UTC |
|---|
| Update Date | 2016-11-09 01:17:50 UTC |
|---|
| Accession Number | CHEM024184 |
|---|
| Identification |
|---|
| Common Name | Austalide K |
|---|
| Class | Small Molecule |
|---|
| Description | Austalide K is a mycotoxin of the food storage mould (Aspergillus ustus |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | Not Available |
|---|
| Chemical Formula | C25H32O5 |
|---|
| Average Molecular Mass | 412.519 g/mol |
|---|
| Monoisotopic Mass | 412.225 g/mol |
|---|
| CAS Registry Number | 87833-53-2 |
|---|
| IUPAC Name | 10-methoxy-1,4,14,18,18-pentamethyl-2,7-dioxapentacyclo[11.8.0.0³,¹¹.0⁵,⁹.0¹⁴,¹⁹]henicosa-3(11),4,9-triene-8,17-dione |
|---|
| Traditional Name | 10-methoxy-1,4,14,18,18-pentamethyl-2,7-dioxapentacyclo[11.8.0.0³,¹¹.0⁵,⁹.0¹⁴,¹⁹]henicosa-3(11),4,9-triene-8,17-dione |
|---|
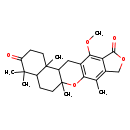
| SMILES | COC1=C2C(=O)OCC2=C(C)C2=C1CC1C(C)(CCC3C(C)(C)C(=O)CCC13C)O2 |
|---|
| InChI Identifier | InChI=1S/C25H32O5/c1-13-15-12-29-22(27)19(15)21(28-6)14-11-17-24(4)9-8-18(26)23(2,3)16(24)7-10-25(17,5)30-20(13)14/h16-17H,7-12H2,1-6H3 |
|---|
| InChI Key | JCUWKPURUZEBFJ-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as isoflavanols. These are polycyclic compounds containing a hydroxylated isoflavan skeleton. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Isoflavonoids |
|---|
| Sub Class | Isoflavans |
|---|
| Direct Parent | Isoflavanols |
|---|
| Alternative Parents | |
|---|
| Substituents | - Isoflavanol
- Dibenzopyran
- Xanthene
- Naphthopyran
- Isobenzofuranone
- Chromane
- Benzopyran
- Naphthalene
- 1-benzopyran
- Phthalide
- Isocoumaran
- Anisole
- Alkyl aryl ether
- Benzenoid
- Pyran
- Cyclic ketone
- Lactone
- Ketone
- Carboxylic acid ester
- Monocarboxylic acid or derivatives
- Ether
- Organoheterocyclic compound
- Carboxylic acid derivative
- Oxacycle
- Organic oxygen compound
- Carbonyl group
- Hydrocarbon derivative
- Organic oxide
- Organooxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-02a2-0309000000-9556b362cd30533f8b7c | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-08fr-0093500000-2b97cb428e1ddc6efc1d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4r-0494100000-36911c6640dacb8a6d54 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0bu0-0942000000-7146aa4735f94e41cf05 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0002900000-94c8964a0d063116030e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-1249700000-7396bbfd7a29561cb44f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-004s-1937000000-52d00a3d0afe7973d041 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0002900000-1e9d954882c5b2580cd3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03dj-2169300000-ae2c073c0ac1e66f1bff | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-01ot-4097100000-aa3cc4d3e6e150992091 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0000900000-612dcdef2b32320e3ae9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-0003900000-cf03a28d5f500b351923 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-1469500000-4d4c290882dc09a87011 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0030157 |
|---|
| FooDB ID | FDB001966 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 26503486 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 13942822 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|