| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:29:39 UTC |
|---|
| Update Date | 2016-11-09 01:17:49 UTC |
|---|
| Accession Number | CHEM024087 |
|---|
| Identification |
|---|
| Common Name | Oryzalexin D |
|---|
| Class | Small Molecule |
|---|
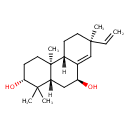
| Description | A tricyclic diterpenoid that is ent-sandaracopimaradiene bearing two additional hydroxy substituents at the 3alpha- and 7beta-positions. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3alpha,7beta-Dihydroxy-ent-sandaracopimaradiene | ChEBI | | ent-Sandaracopimaradien-3alpha,7beta-diol | ChEBI | | 3a,7b-Dihydroxy-ent-sandaracopimaradiene | Generator | | 3Α,7β-dihydroxy-ent-sandaracopimaradiene | Generator | | ent-Sandaracopimaradien-3a,7b-diol | Generator | | ent-Sandaracopimaradien-3α,7β-diol | Generator |
|
|---|
| Chemical Formula | C20H32O2 |
|---|
| Average Molecular Mass | 304.467 g/mol |
|---|
| Monoisotopic Mass | 304.240 g/mol |
|---|
| CAS Registry Number | 110268-98-9 |
|---|
| IUPAC Name | (2R,4aR,4bS,7S,9S,10aS)-7-ethenyl-1,1,4a,7-tetramethyl-1,2,3,4,4a,4b,5,6,7,9,10,10a-dodecahydrophenanthrene-2,9-diol |
|---|
| Traditional Name | (2R,4aR,4bS,7S,9S,10aS)-7-ethenyl-1,1,4a,7-tetramethyl-3,4,4b,5,6,9,10,10a-octahydro-2H-phenanthrene-2,9-diol |
|---|
| SMILES | [H][C@@]12CC[C@@](C)(C=C)C=C1[C@@H](O)C[C@]1([H])C(C)(C)[C@H](O)CC[C@@]21C |
|---|
| InChI Identifier | InChI=1S/C20H32O2/c1-6-19(4)9-7-14-13(12-19)15(21)11-16-18(2,3)17(22)8-10-20(14,16)5/h6,12,14-17,21-22H,1,7-11H2,2-5H3/t14-,15+,16-,17-,19-,20+/m1/s1 |
|---|
| InChI Key | HRWWBCRSPUEXDM-XTMWUNHTSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as diterpenoids. These are terpene compounds formed by four isoprene units. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Diterpenoids |
|---|
| Direct Parent | Diterpenoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Pimarane diterpenoid
- Diterpenoid
- Phenanthrene
- Hydrophenanthrene
- Cyclic alcohol
- Secondary alcohol
- Organic oxygen compound
- Hydrocarbon derivative
- Organooxygen compound
- Alcohol
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-052r-0092000000-a7a2782a0f4ee2bcf61b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-05n0-3190000000-dbd7ad89ccbe95e093cb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0zfr-9070000000-a7a4ce9e18c9b8dbe180 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0049000000-c414589d392935e0b2e3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udr-0097000000-97ba0d0d4eec8f7cada5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0fki-3090000000-8560cdbe6ba928c3c43d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0079000000-efb679c558be1586a5a6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a5i-3491000000-87a93813ec3bae71c0bb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-000m-9820000000-6c3990d393701d6c22aa | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0009000000-f9d10b76ade80bd401ad | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-0009000000-f9d10b76ade80bd401ad | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0udj-0089000000-7c46de12c936bd072d45 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0302102 |
|---|
| FooDB ID | FDB001859 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00034092 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | CPD-16612 |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 31150507 |
|---|
| ChEBI ID | 78256 |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|