| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:26:49 UTC |
|---|
| Update Date | 2016-11-09 01:17:49 UTC |
|---|
| Accession Number | CHEM024030 |
|---|
| Identification |
|---|
| Common Name | Cichorioside G |
|---|
| Class | Small Molecule |
|---|
| Description | Not Available |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
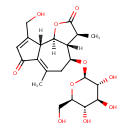
| Chemical Structure | |
|---|
| Synonyms | Not Available |
|---|
| Chemical Formula | C21H28O10 |
|---|
| Average Molecular Mass | 440.441 g/mol |
|---|
| Monoisotopic Mass | 440.168 g/mol |
|---|
| CAS Registry Number | 1086489-75-9 |
|---|
| IUPAC Name | (3S,3aR,4S,9aS,9bR)-9-(hydroxymethyl)-3,6-dimethyl-4-{[(2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}-2H,3H,3aH,4H,5H,7H,9aH,9bH-azuleno[4,5-b]furan-2,7-dione |
|---|
| Traditional Name | (3S,3aR,4S,9aS,9bR)-9-(hydroxymethyl)-3,6-dimethyl-4-{[(2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}-3H,3aH,4H,5H,9aH,9bH-azuleno[4,5-b]furan-2,7-dione |
|---|
| SMILES | [H][C@]12OC(=O)[C@@H](C)[C@]1([H])[C@H](CC(C)=C1C(=O)C=C(CO)[C@]21[H])O[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O |
|---|
| InChI Identifier | InChI=1S/C21H28O10/c1-7-3-11(29-21-18(27)17(26)16(25)12(6-23)30-21)14-8(2)20(28)31-19(14)15-9(5-22)4-10(24)13(7)15/h4,8,11-12,14-19,21-23,25-27H,3,5-6H2,1-2H3/t8-,11-,12+,14+,15-,16+,17-,18+,19-,21+/m0/s1 |
|---|
| InChI Key | NUAIXFCSBZGUAF-PNSMZNPGSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as o-glycosyl compounds. These are glycoside in which a sugar group is bonded through one carbon to another group via a O-glycosidic bond. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbohydrates and carbohydrate conjugates |
|---|
| Direct Parent | O-glycosyl compounds |
|---|
| Alternative Parents | |
|---|
| Substituents | - Hexose monosaccharide
- O-glycosyl compound
- Gamma butyrolactone
- Monosaccharide
- Oxane
- Tetrahydrofuran
- Carboxylic acid ester
- Ketone
- Lactone
- Secondary alcohol
- Oxacycle
- Monocarboxylic acid or derivatives
- Acetal
- Carboxylic acid derivative
- Organoheterocyclic compound
- Polyol
- Alcohol
- Hydrocarbon derivative
- Organic oxide
- Carbonyl group
- Primary alcohol
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03ml-0091600000-69e6d641b6e6521defb9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03di-0290100000-b3f2271491ed3f3fdefd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-03dr-0490000000-d39fcb90c207a7bab1eb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0550-0173900000-bd897299639b96226e1a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-056r-0191100000-13922c5ae4f4b3d0bb75 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-010r-1090000000-7eb910dca8c542965461 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0090000000-d52acc429011baa533bb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03di-0190000000-3b1be63cbe47128dfe35 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-07w9-1290000000-2dc9944a6bd16e7b0cc8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0021900000-41f3638940433c9eac7c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0550-9176600000-75dc2c46f28670291779 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a6s-2091000000-5b856660bb1467b9a237 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0302053 |
|---|
| FooDB ID | FDB001799 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00029953 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 59696259 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 102476814 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|