| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:24:48 UTC |
|---|
| Update Date | 2016-11-09 01:17:48 UTC |
|---|
| Accession Number | CHEM023991 |
|---|
| Identification |
|---|
| Common Name | Apo-10'-zeaxanthinal |
|---|
| Class | Small Molecule |
|---|
| Description | 3-Hydroxy-10'-apo-b,y-carotenal is found in citrus. 3-Hydroxy-10'-apo-b,y-carotenal isisolated from Sinton citrangequat. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
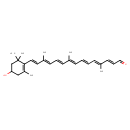
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3-Hydroxy-10'-apo-b,y-carotenal, 9ci | HMDB | | 3-Hydroxy-b-apo-10'-carotenal | HMDB | | apo-3-Zeaxanthinal | HMDB |
|
|---|
| Chemical Formula | C27H36O2 |
|---|
| Average Molecular Mass | 392.574 g/mol |
|---|
| Monoisotopic Mass | 392.272 g/mol |
|---|
| CAS Registry Number | 62742-01-2 |
|---|
| IUPAC Name | (2E,4E,6E,8E,10E,12E,14E)-15-(4-hydroxy-2,6,6-trimethylcyclohex-1-en-1-yl)-4,9,13-trimethylpentadeca-2,4,6,8,10,12,14-heptaenal |
|---|
| Traditional Name | (2E,4E,6E,8E,10E,12E,14E)-15-(4-hydroxy-2,6,6-trimethylcyclohex-1-en-1-yl)-4,9,13-trimethylpentadeca-2,4,6,8,10,12,14-heptaenal |
|---|
| SMILES | C\C(\C=C\C=O)=C/C=C/C=C(\C)/C=C/C=C(\C)/C=C/C1=C(C)CC(O)CC1(C)C |
|---|
| InChI Identifier | InChI=1S/C27H36O2/c1-21(11-7-8-12-22(2)15-10-18-28)13-9-14-23(3)16-17-26-24(4)19-25(29)20-27(26,5)6/h7-18,25,29H,19-20H2,1-6H3/b8-7+,13-9+,15-10+,17-16+,21-11+,22-12+,23-14+ |
|---|
| InChI Key | HQPQTQQUHPFUOA-JNOJMRGUSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as sesterterpenoids. These are terpenes composed of five consecutive isoprene units. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Sesterterpenoids |
|---|
| Direct Parent | Sesterterpenoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Sesterterpenoid
- Enal
- Alpha,beta-unsaturated aldehyde
- Secondary alcohol
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Aldehyde
- Alcohol
- Aliphatic homomonocyclic compound
|
|---|
| Molecular Framework | Aliphatic homomonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-004i-1009000000-9ecf83e646d5fe1efd4a | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-0002-6104900000-8c0a30ee23f38d21bcd4 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004l-0219000000-9c0d84576a58f25f3a32 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-003i-3954000000-0ca3e9f5eaa0bf613c9d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0059-9867000000-4dfb9be65b13b71ce0f6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0009000000-556ae78ec917f2292e29 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-006x-0009000000-543471504caa5110115e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0007-4239000000-c30dc096a2c19c6d7383 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-0009000000-5d01424d64d86a92b426 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-01ow-0109000000-231cd63302db39160576 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-001j-1639000000-7ea6ef06ed51ebae8d62 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000f-0239000000-6c50c9d1cd24ab0560e5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0005-1339000000-ae428930081c746ebcc6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0002-2930000000-74dc53f773decc804300 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0039019 |
|---|
| FooDB ID | FDB018514 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 35014718 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 87443544 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|