| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:24:44 UTC |
|---|
| Update Date | 2016-11-09 01:17:48 UTC |
|---|
| Accession Number | CHEM023989 |
|---|
| Identification |
|---|
| Common Name | Jacquinelin |
|---|
| Class | Small Molecule |
|---|
| Description | A sesquiterpene lactone obtained by formal hydrogenation across the 11,13-double bond of 8-deoxylactucin. Found in chicory |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
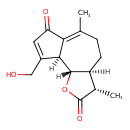
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 11,13-Dihydro-8-deoxylactucin | ChEBI | | Jacquilenin | ChEBI | | 11b,13-Dihydro-8-deoxylactucin | Generator | | 11Β,13-dihydro-8-deoxylactucin | Generator |
|
|---|
| Chemical Formula | C15H18O4 |
|---|
| Average Molecular Mass | 262.301 g/mol |
|---|
| Monoisotopic Mass | 262.121 g/mol |
|---|
| CAS Registry Number | 7726-34-3 |
|---|
| IUPAC Name | (3S,3aS,9aS,9bS)-9-(hydroxymethyl)-3,6-dimethyl-2H,3H,3aH,4H,5H,7H,9aH,9bH-azuleno[4,5-b]furan-2,7-dione |
|---|
| Traditional Name | (3S,3aS,9aS,9bS)-9-(hydroxymethyl)-3,6-dimethyl-3H,3aH,4H,5H,9aH,9bH-azuleno[4,5-b]furan-2,7-dione |
|---|
| SMILES | [H][C@@]12CCC(C)=C3C(=O)C=C(CO)[C@]3([H])[C@@]1([H])OC(=O)[C@H]2C |
|---|
| InChI Identifier | InChI=1S/C15H18O4/c1-7-3-4-10-8(2)15(18)19-14(10)13-9(6-16)5-11(17)12(7)13/h5,8,10,13-14,16H,3-4,6H2,1-2H3/t8-,10-,13-,14-/m0/s1 |
|---|
| InChI Key | SNIFBMIPCYBVSS-LMVZTGKYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as gamma butyrolactones. Gamma butyrolactones are compounds containing a gamma butyrolactone moiety, which consists of an aliphatic five-member ring with four carbon atoms, one oxygen atom, and bears a ketone group on the carbon adjacent to the oxygen atom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Lactones |
|---|
| Sub Class | Gamma butyrolactones |
|---|
| Direct Parent | Gamma butyrolactones |
|---|
| Alternative Parents | |
|---|
| Substituents | - Gamma butyrolactone
- Tetrahydrofuran
- Ketone
- Carboxylic acid ester
- Oxacycle
- Monocarboxylic acid or derivatives
- Carboxylic acid derivative
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Primary alcohol
- Organooxygen compound
- Carbonyl group
- Alcohol
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-01ot-0190000000-ac66bdbb74893249fd0d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00ba-0960000000-ef9be809a79767b25af3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0umi-7910000000-6c59802ec51c65674d39 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0090000000-91777ed03c63829497bb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03yl-0190000000-1e18d5f2444266325897 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0h90-2910000000-2e81217885a9fdbbf0ae | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0090000000-9f75870e3f0a622d8ae4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03ds-0290000000-a0d67b88ac62ef082635 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-000l-0950000000-992f46af351f1dfdd1de | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0090000000-92a5419893bc0c0dd3ae | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-0090000000-fbe47b2803da998e469d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-0090000000-ddb3f9db5004feca1b68 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0302021 |
|---|
| FooDB ID | FDB001755 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 23326587 |
|---|
| ChEBI ID | 90279 |
|---|
| PubChem Compound ID | 14163574 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|