| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:24:17 UTC |
|---|
| Update Date | 2016-11-09 01:17:48 UTC |
|---|
| Accession Number | CHEM023975 |
|---|
| Identification |
|---|
| Common Name | Afzelechin-(4alpha->8)-pelargonidin 3-O-beta-glucopyranoside |
|---|
| Class | Small Molecule |
|---|
| Description | Afzelechin-(4alpha->8)-pelargonidin 3-o-beta-glucopyranoside is practically insoluble (in water) and a very weakly acidic compound (based on its pKa). Afzelechin-(4alpha->8)-pelargonidin 3-o-beta-glucopyranoside can be found in strawberry, which makes afzelechin-(4alpha->8)-pelargonidin 3-o-beta-glucopyranoside a potential biomarker for the consumption of this food product. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
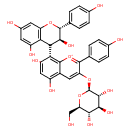
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Afzelechin-(4a->8)-pelargonidin 3-O-b-glucopyranoside | Generator | | Afzelechin-(4α->8)-pelargonidin 3-O-β-glucopyranoside | Generator |
|
|---|
| Chemical Formula | C36H33O15 |
|---|
| Average Molecular Mass | 705.638 g/mol |
|---|
| Monoisotopic Mass | 705.182 g/mol |
|---|
| CAS Registry Number | 753008-66-1 |
|---|
| IUPAC Name | 5,7-dihydroxy-2-(4-hydroxyphenyl)-8-[(2R,3S)-3,5,7-trihydroxy-2-(4-hydroxyphenyl)-3,4-dihydro-2H-1-benzopyran-4-yl]-3-{[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}-1λ⁴-chromen-1-ylium |
|---|
| Traditional Name | 5,7-dihydroxy-2-(4-hydroxyphenyl)-8-[(2R,3S)-3,5,7-trihydroxy-2-(4-hydroxyphenyl)-3,4-dihydro-2H-1-benzopyran-4-yl]-3-{[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}-1λ⁴-chromen-1-ylium |
|---|
| SMILES | OC[C@H]1O[C@@H](OC2=CC3=C(O)C=C(O)C(C4[C@H](O)[C@H](OC5=C4C(O)=CC(O)=C5)C4=CC=C(O)C=C4)=C3[O+]=C2C2=CC=C(O)C=C2)[C@H](O)[C@@H](O)[C@@H]1O |
|---|
| InChI Identifier | InChI=1S/C36H32O15/c37-13-25-29(44)31(46)32(47)36(50-25)49-24-11-19-20(41)12-22(43)27(35(19)51-33(24)14-1-5-16(38)6-2-14)28-26-21(42)9-18(40)10-23(26)48-34(30(28)45)15-3-7-17(39)8-4-15/h1-12,25,28-32,34,36-37,44-47H,13H2,(H5-,38,39,40,41,42,43)/p+1/t25-,28?,29-,30+,31+,32-,34-,36-/m1/s1 |
|---|
| InChI Key | UCHHYHHTRCIREP-IEPQWAIVSA-O |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as iridoids and derivatives. These are monoterpenes containing a skeleton structurally characterized by the presence of a cylopentane fused to a pyran ( forming a 4,7-dimethylcyclopenta[c]pyran), or a derivative where the pentane moiety is open. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Monoterpenoids |
|---|
| Direct Parent | Iridoids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Monocyclic monoterpenoid
- 11-noriridane monoterpenoid
- Cyclic ketone
- Ketone
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Carboxylic acid derivative
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Aliphatic homomonocyclic compound
|
|---|
| Molecular Framework | Aliphatic homomonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0100000900-1da6dbb852efc848da97 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014j-1400024900-fa4d13c1d742ea9a87aa | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-03dj-5900111100-3706bdcd57ae799a2954 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-2300000900-1b3f5aea0ff7c248baab | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0w29-6800000900-463d98439c9390512e1e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-006x-9400000000-7502f75023aba234be18 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | FDB001740 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|