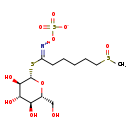
| (2R,3S,4S,5R,6S)-2-(Hydroxymethyl)-6-({6-methanesulphinyl-1-[(sulphonatooxy)imino]hexyl}sulphanyl)oxane-3,4,5-triol | Generator |
| 5-Methylsulfinylpentyl glucosinolate | ChEBI |
| beta-D-Glucopyranose, 1-thio-, 1-(6-(methylsulfinyl)-N-(sulfooxy)hexanimidate) | ChEBI |
| 5-Methylsulfinylpentyl glucosinolic acid | Generator |
| 5-Methylsulphinylpentyl glucosinolate | Generator |
| 5-Methylsulphinylpentyl glucosinolic acid | Generator |
| b-D-Glucopyranose, 1-thio-, 1-(6-(methylsulfinyl)-N-(sulfooxy)hexanimidate) | Generator |
| b-D-Glucopyranose, 1-thio-, 1-(6-(methylsulfinyl)-N-(sulfooxy)hexanimidic acid) | Generator |
| b-D-Glucopyranose, 1-thio-, 1-(6-(methylsulphinyl)-N-(sulphooxy)hexanimidate) | Generator |
| b-D-Glucopyranose, 1-thio-, 1-(6-(methylsulphinyl)-N-(sulphooxy)hexanimidic acid) | Generator |
| beta-D-Glucopyranose, 1-thio-, 1-(6-(methylsulfinyl)-N-(sulfooxy)hexanimidic acid) | Generator |
| beta-D-Glucopyranose, 1-thio-, 1-(6-(methylsulphinyl)-N-(sulphooxy)hexanimidate) | Generator |
| beta-D-Glucopyranose, 1-thio-, 1-(6-(methylsulphinyl)-N-(sulphooxy)hexanimidic acid) | Generator |
| β-D-glucopyranose, 1-thio-, 1-(6-(methylsulfinyl)-N-(sulfooxy)hexanimidate) | Generator |
| β-D-glucopyranose, 1-thio-, 1-(6-(methylsulfinyl)-N-(sulfooxy)hexanimidic acid) | Generator |
| β-D-glucopyranose, 1-thio-, 1-(6-(methylsulphinyl)-N-(sulphooxy)hexanimidate) | Generator |
| β-D-glucopyranose, 1-thio-, 1-(6-(methylsulphinyl)-N-(sulphooxy)hexanimidic acid) | Generator |