| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:14:26 UTC |
|---|
| Update Date | 2016-11-09 01:17:45 UTC |
|---|
| Accession Number | CHEM023701 |
|---|
| Identification |
|---|
| Common Name | 5-Hydroxyglucobrassicin |
|---|
| Class | Small Molecule |
|---|
| Description | Not Available |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
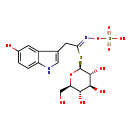
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| {[(Z)-[2-(5-hydroxy-1H-indol-3-yl)-1-{[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]sulfanyl}ethylidene]amino]oxy}sulfonate | Generator | | {[(Z)-[2-(5-hydroxy-1H-indol-3-yl)-1-{[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]sulphanyl}ethylidene]amino]oxy}sulphonate | Generator | | {[(Z)-[2-(5-hydroxy-1H-indol-3-yl)-1-{[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]sulphanyl}ethylidene]amino]oxy}sulphonic acid | Generator |
|
|---|
| Chemical Formula | C16H20N2O10S2 |
|---|
| Average Molecular Mass | 464.467 g/mol |
|---|
| Monoisotopic Mass | 464.056 g/mol |
|---|
| CAS Registry Number | 87592-99-2 |
|---|
| IUPAC Name | {[(Z)-[2-(5-hydroxy-1H-indol-3-yl)-1-{[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]sulfanyl}ethylidene]amino]oxy}sulfonic acid |
|---|
| Traditional Name | [(Z)-[2-(5-hydroxy-1H-indol-3-yl)-1-{[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]sulfanyl}ethylidene]amino]oxysulfonic acid |
|---|
| SMILES | OC[C@H]1O[C@@H](S\C(=N/OS(O)(=O)=O)CC2=CNC3=CC=C(O)C=C23)[C@H](O)[C@@H](O)[C@@H]1O |
|---|
| InChI Identifier | InChI=1S/C16H20N2O10S2/c19-6-11-13(21)14(22)15(23)16(27-11)29-12(18-28-30(24,25)26)3-7-5-17-10-2-1-8(20)4-9(7)10/h1-2,4-5,11,13-17,19-23H,3,6H2,(H,24,25,26)/b18-12-/t11-,13-,14+,15-,16+/m1/s1 |
|---|
| InChI Key | SNGNIBNFTIUEHD-PIAXYHQTSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as alkylglucosinolates. These are organic compounds containing a glucosinolate moiety that carries an alkyl chain. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbohydrates and carbohydrate conjugates |
|---|
| Direct Parent | Alkylglucosinolates |
|---|
| Alternative Parents | |
|---|
| Substituents | - Alkylglucosinolate
- Glycosyl compound
- S-glycosyl compound
- Hydroxyindole
- 3-alkylindole
- Indole
- Indole or derivatives
- 1-hydroxy-2-unsubstituted benzenoid
- Oxane
- Benzenoid
- Substituted pyrrole
- Monothioacetal
- Heteroaromatic compound
- Organic sulfuric acid or derivatives
- Pyrrole
- Secondary alcohol
- Oxacycle
- Azacycle
- Polyol
- Organoheterocyclic compound
- Sulfenyl compound
- Organosulfur compound
- Organonitrogen compound
- Alcohol
- Hydrocarbon derivative
- Organic nitrogen compound
- Primary alcohol
- Organopnictogen compound
- Organic oxide
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-0914700000-2a03cdc22760b989b719 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-1159-0948000000-02ee3e810d572b7c20e6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0002-5900000000-8b74cdd599ad9ab5f16e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-3239100000-ae394220720516afb700 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-014i-7960000000-d17a6e71d5058d880f4b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-030u-6940000000-760b336c2edc86b35617 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0000900000-9411b93aecef03be4230 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00mk-0105900000-c649169dc3b2404ef904 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-052b-1983200000-ab6666eada7b4b20cff3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0900600000-5f5ca95d94ae70790b01 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0ik9-1319200000-da90ae7e6e31f74f1b6a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0ue9-0900000000-214df2fb8a789428a4c3 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0301786 |
|---|
| FooDB ID | FDB001409 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 59696183 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|